Hydatid Cyst of The Orbit: A Systematic Review with Meta-Data
Abstarct
Introduction
Orbital hydatid cysts (HCs) constitute less than 1% of all cases of hydatidosis, yet their occurrence is often linked to severe visual complications. This study presents a systematic review of reported cases of orbital HCs.
Methods
A systematic review of the published studies of orbital HCs was conducted, the studies that met the following criteria were included: 1) The presence of the infection was confirmed through diagnostic methods, surgical findings, or histopathology. 2) The study provided a detailed case presentation.
Results
Thirty-two studies (56 cases) met the inclusion criteria. Ten patients were from Afghanistan (17.9%). There was no gender predilection, the distribution was almost equal. The ages ranged from three to 80 years old. The most common symptoms that the patients presented with were proptosis of the affected eye (98.2%) and visual impairment (64.3%). The therapeutic approach of orbital HC was primarily surgical removal of the cyst accompanied by anthelmintic drugs in 41 (73.2%) cases. Concurrent HC was reported in two cases (3.6%), and recurrence with subsequent recovery was reported in four (7.1%) cases.
Conclusion
Orbital HC is a rare condition, primarily diagnosed using MRI, with surgery as the definitive treatment. Concurrent hydatidosis increases the risk of recurrence, requiring thorough and ongoing follow-up.
Introduction
Hydatidosis or hydatid cyst (HC) is a commonly recognized zoonotic disease caused by the larval form of the tapeworm Echinococcus granulosus. Humans act as intermediate hosts for this parasite, acquiring infection through direct contact with definitive hosts (e.g., sheep, goats, cattle, dogs) or consuming contaminated food or water. [1].
The global incidence of hydatidosis varies, with higher rates observed in regions where livestock farming is widespread. Key risk factors for contracting hydatidosis include close contact with dogs, livestock-related activities, and residence in areas where the disease is endemic. These cysts typically occur in the liver (50-70%) and lungs (20-30%). The global burden of HC is significant, with an estimated 2 to 3 million cases reported worldwide [2]. However, orbital HC is uncommon, representing less than 1% of all cases, accounting for 19.8% in endemic countries [3].
The World Health Organization (WHO) has classified Echinococcosis as one of the 20 neglected tropical diseases that pose significant public health concerns. To ensure consistent global monitoring, the WHO Informal Working Group on echinococcosis has categorized cysts of echinococcosis into five distinct types, grouped into three main categories. Specifically, CE1 and CE2 are indicative of active infection, CE3 represents an intermediate stage, while CE4 and CE5 are associated with inactive cysts [4].
In endemic regions, environmental and climatic conditions play a crucial role in the survival of parasite eggs and the living conditions of livestock and stray dogs. For example, Echinococcus granulosus eggs remain viable in water and damp sand for up to three weeks at 30°C, 4.5 weeks at 10–21°C, and 32 weeks at 6°C. They can also survive for several months in green pastures and gardens [4]. Although the WHO classifies hydatidosis as a neglected disease, it continues to be a significant public health concern due to its status as the second most impactful foodborne parasitic disease, its endemic presence in certain regions, and its potential to cause substantial morbidity. The WHO prioritizes the control and prevention of hydatidosis, particularly given its impact on human health, animals, and the food supply chain.
Orbital HC, although rare, is often linked to severe visual complications. As of the date of the current review, the available literature on orbital HC primarily consists of case reports and case series, with no reviews currently available. This study aims to provide and analyze a collection of data through a systematic review and a meta-data presentation.
Methods
Study design
This systematic review was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [5].
Data sources and search strategy
A systematic review of the published studies of orbital HCs was conducted using Google Scholar and PubMed. Boolean operators (OR/AND) were used to refine the results. The keywords that were used in the search included: (eye OR orbital OR intraorbital OR ocular) AND (hydatid OR echinococcosis OR hydatidosis).
Eligibility criteria
Studies in languages other than English, as well as those not related to humans, were excluded either before or during the initial screening process. All studies on orbital HCs that met the following criteria were included: 1) The presence of the infection was confirmed through diagnostic methods, surgical findings, or histopathology. 2) The case presentation was detailed in the study. Studies published in non-peer-reviewed journals [6] or those failing to meet inclusion criteria were excluded.
Selection and extraction of data
The titles and abstracts of identified studies were first screened, followed by a thorough full-text review to assess eligibility. Key data were extracted from the included studies, including study design, country of origin, patient demographics (age, gender, residence), symptoms, history of HC, serological tests, diagnosis, management strategies, follow-up details, and recurrence rates.
Data analysis
Microsoft Excel (2019) was used to gather and organize the extracted data, while the Statistical Package for Social Sciences (SPSS) version 27.0 was utilized for data analysis (descriptive statistics). The findings were displayed as frequencies, percentages, ranges, and means with standard deviations.
Results
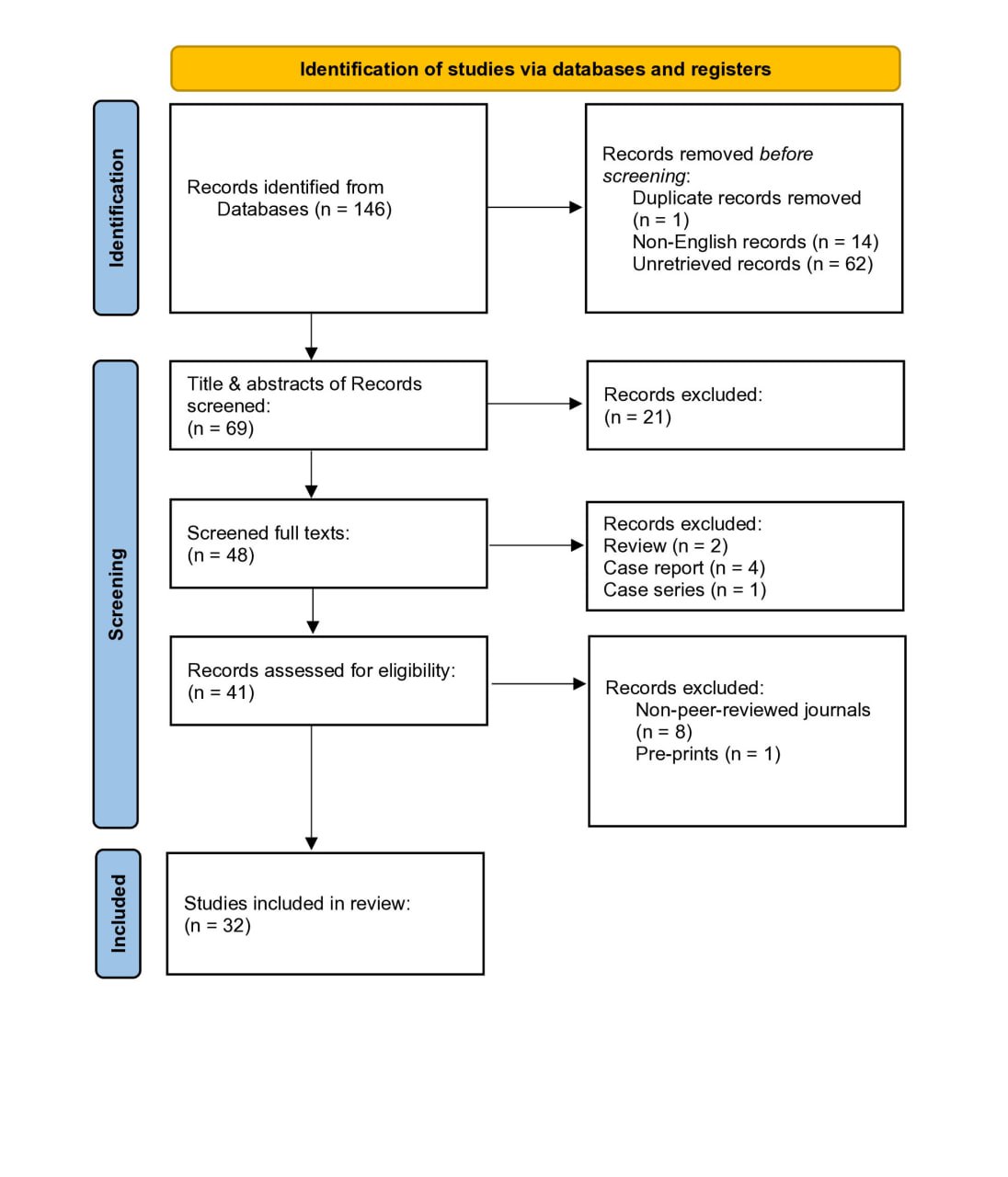
A total of 146 studies were retrieved. One was excluded as a duplicate, 14 were non-English, and 62 were unretrievable. After title and abstract screening, 21 studies did not meet the inclusion criteria. The remaining 48 underwent full-text review, with seven more excluded. Of the 41 studies assessed for eligibility, nine were excluded for being from non-peer-reviewed journals or preprints. Ultimately, 32 studies [3,7-37] (56 cases) met the inclusion criteria (Figure 1).
Of the included studies [3,7-37], 28 (87.5%) were case reports, while the remaining 4 (12.5%) were case series (Table 1). The highest number of patients were from Afghanistan (10, 17.9%), followed by India (8, 14.3%), Azerbaijan (8, 14.3%), and Morocco and Turkey (6 each, 10.7%). Patient ages spanned from 3 to 80 years, with a mean age of 27.45 ± 19.57 years. The majority of the cases occurred between the first and fifth decades of life (47, 83.9%). The right side was affected in 33 (58.9%) cases and there were no cases with bilateral HC. Sixteen patients (28.6%) were from rural areas, and 13 (23.2%) reported contact with dogs, sheep, or other cattle (Table 2).
|
Author |
Type of study |
Country of the patients |
N. of Patients |
Age |
Sex |
Symptoms |
Affected side |
Cyst Size (Cm) |
Surgical approach |
Cyst removal approach |
Adjuvant therapy |
Outcome |
Follow up (months) |
|
|
Abouassi et al. [3] |
Case Report |
Syria |
1 |
21 |
F |
Proptosis & visual impairment |
Right |
4.2 |
Fronto-orbitozy-gomatic orbitotomy |
Cystectomy |
Albendazole |
Recovered |
3 |
|
|
Ilhami et al. [7] |
Case series |
Morocco |
3 |
13 |
F |
Proptosis & decreased visual acuity |
Right |
4.2 |
Internal paracanthal orbitotomy |
Cystectomy |
Albendazole |
Recovered |
N/A |
|
|
67 |
F |
Proptosis, pain, headache & chemosis |
Left |
3.5 |
Superolateral orbitotomy |
Enucleation cystectomy |
Albendazole |
Recovered |
N/A |
|||||
|
43 |
F |
Proptosis |
Left |
2.9 |
Internal paracanthal orbitotomy |
Enucleation cystectomy |
Albendazole |
Recovered |
N/A |
|||||
Alabdullah et al. [8] |
Case Report |
Syria |
1 |
10 |
M |
Proptosis, diplopia & decreased vision |
Left |
2.7 |
Subperiosteal orbitotomy |
Lynch method |
Albendazole |
Recovered |
N/A |
|
|
Khan et al. [9] |
Case Series |
Pakistan |
11 |
15 |
F |
Proptosis & visual impairment |
Left |
N/A |
Orbitotomy |
Unspecified |
Albendazole |
Recovered |
* |
|
|
15 |
M |
Proptosis & visual impairment |
Right |
N/A |
Orbitotomy |
Unspecified |
Albendazole |
Recovered |
* |
|||||
|
3 |
F |
Proptosis & visual impairment |
Left |
N/A |
Orbitotomy |
Unspecified |
Albendazole |
Recovered |
* |
|||||
|
17 |
F |
Proptosis& visual impairment |
Left |
N/A |
Orbitotomy |
Unspecified |
Albendazole |
Recovered |
* |
|||||
|
28 |
F |
Proptosis & visual impairment |
Right |
N/A |
Orbitotomy |
Unspecified |
Albendazole |
Recovered |
* |
|||||
|
19 |
M |
Proptosis & visual impairment |
Left |
N/A |
Orbitotomy |
Unspecified |
Albendazole |
Recovered |
* |
|||||
|
20 |
F |
Proptosis & visual impairment |
Left |
N/A |
Orbitotomy |
Unspecified |
Albendazole |
Recovered |
* |
|||||
|
6 |
M |
Proptosis & visual impairment |
Right |
N/A |
Orbitotomy |
Unspecified |
Albendazole |
Recovered |
* |
|||||
|
6 |
M |
Proptosis & visual impairment |
Right |
N/A |
Orbitotomy |
Unspecified |
Albendazole |
Recovered |
* |
|||||
|
5 |
M |
Proptosis & visual impairment |
Left |
N/A |
Orbitotomy |
Unspecified |
Albendazole |
Recovered |
* |
|||||
|
65 |
M |
Proptosis & visual impairment |
Left |
N/A |
Orbitotomy |
Unspecified |
Albendazole |
Recovered |
* |
|||||
|
Bamashmus et al. [18] |
Case report |
Yemen |
1 |
58 |
M |
Proptosis, impaired vision & chemosis |
Right |
N/A |
Transconjuctival & lateral orbitotomy |
PAIR method |
Mebendazole |
Recovered |
N/A |
|
|
Assimakopoulos et al. [19] |
Case report |
Greece |
1 |
31 |
F |
Proptosis & impaired vision |
Left |
N/A |
Lateral orbitotomy |
Modified cystectomy |
Albendazole |
Recovered |
3 |
|
|
Berradi et al. [20] |
Case report |
Morocco |
1 |
46 |
M |
Proptosis |
Left |
4.2 |
Unspecified |
Modified PAIR method |
None |
Recovered |
3 |
|
|
Chitra et al. [21] |
Case report |
Morocco |
1 |
3 |
F |
Proptosis & impaired vision |
Left |
2.8 |
Extradural frontal orbitotomy |
Barrett’s technique |
Albendazole |
Recovered |
24 |
|
|
Elkrimi et al. [22] |
Case Report |
Morocco |
1 |
5 |
M |
Proptosis |
Left |
3.1 |
Combined approach (endoscopy & supraorbital incision) |
Partial cystectomy |
Albendazole |
Recovered |
6 |
|
|
Hosaini et al. [23] |
Case report |
Afghanistan |
1 |
8 |
M |
Proptosis, chemosis, reduced vision & headache |
Right |
5 |
Transconjuctival orbitotomy |
Modified cystectomy |
Albendazole |
Recovered |
N/A |
|
|
Jaffar et al. [24] |
Case report |
Pakistan |
1 |
27 |
M |
Proptosis, visual impairment, reduced ocular motion & discharge |
Left |
5 |
Unspecified |
Unspecified |
None |
Recovered |
N/A |
|
|
Kars et al. [25] |
Case report |
Turkey |
2 |
7 |
M |
Proptosis & impaired vision |
Left |
N/A |
Transcranial orbitotomy |
Unspecified |
None |
Had recurrence, recovered after a second surgery |
24 |
|
|
11 |
F |
Proptosis, impaired vision & limited ocular motility |
Right |
N/A |
Transcranial orbitotomy |
Unspecified |
None |
Recovered |
6 |
|||||
|
Das et al. [26] |
Case report |
India |
1 |
52 |
M |
Proptosis |
Left |
4 |
Orbitotomy |
Unspecified |
Albendazole |
N/A |
N/A |
|
|
Motlagh et al. [27] |
Case report |
Iran |
1 |
24 |
M |
Proptosis & diplopia |
Right |
N/A |
Frontotemporal craniotomy & superior orbitotomy |
Partial cystectomy with saline irrigation |
Albendazole, antibiotics & steroid |
Recovered |
N/A |
|
|
Özek et al. [28] |
Case report |
Turkey |
1 |
52 |
F |
Proptosis, visual loss & orbital pain |
Right |
N/A |
Lateral orbitotomy |
Cystectomy with saline irrigation |
Mebendazole |
Recovered |
7 |
|
|
Rajabi et al. [29]
|
Case series
|
Azerbaijan
|
8
|
14 |
M |
Proptosis |
Right |
N/A |
Lateral orbitotomy |
Unspecified |
Albendazole |
Recovered |
** |
|
|
24 |
M |
Proptosis |
Right |
N/A |
Medial orbitotomy |
Unspecified |
Albendazole |
Recovered |
** |
|||||
|
13 |
M |
Proptosis |
Right |
N/A |
Superior orbitotomy |
Unspecified |
Albendazole |
Recovered |
** |
|||||
|
18 |
F |
Proptosis |
Left |
N/A |
Lateral orbitotomy |
Unspecified |
Albendazole |
Recovered |
** |
18 |
||||
|
62 |
F |
Proptosis |
Left |
N/A |
Lateral orbitotomy |
Unspecified |
Albendazole |
Recovered |
** |
62 |
||||
|
33 |
F |
Proptosis |
Right |
N/A |
Lateral orbitotomy |
Unspecified |
Albendazole |
Recovered |
** |
33 |
||||
|
44 |
F |
Proptosis |
Left |
N/A |
Inferior orbitotomy |
Unspecified |
Albendazole |
Recovered |
** |
44 |
||||
|
26 |
M |
Proptosis |
Left |
N/A |
Lateral orbitotomy |
Unspecified |
Albendazole |
Recovered |
** |
26 |
||||
|
Haydar et al. [10] |
Case report |
Afghanistan |
1 |
22 |
M |
Proptosis, decreased vision &pain |
Left |
3.6 |
Inferior transconjunctival orbitotomy |
Aspiration and excision |
Albendazole |
Recovered |
10 |
|
|
Sendul et al. [11] |
Case report |
Turkey |
1 |
24 |
F |
Proptosis & visual impairment |
Right |
2.2 |
Medial transconjonctival orbitotomy |
Cystectomy with aspiration |
Albendazole |
Had recurrence, recovered after a second surgery |
N/A |
|
|
Mathad et al. [12] |
Case Report |
India |
1 |
80 |
F |
Proptosis & visual impairment |
Left |
3 |
Lateral orbitotomy |
Cystectomy |
None |
Recovered |
N/A |
|
|
Öztekin et al. [13] |
Case Report |
Turkey |
1 |
57 |
M |
Proptosis & visual impairment |
Right |
1.5 |
unspecified |
Unspecified |
None |
Recovered |
N/A |
|
|
Kumar et al. [14] |
Case Report |
India |
1 |
47 |
F |
Proptosis. Headache, pain & visual impairment |
Left |
3.7 |
Orbitotomy |
Modified cystectomy |
Albendazole |
Recovered |
12 |
|
|
Debela et al. [15] |
Case Report |
Ethiopia |
1 |
60 |
F |
Proptosis & visual impairment |
Left |
2.6 |
Medial anterior orbitotomy |
Modified cystectomy |
Albendazole |
Recovered |
3 weeks |
|
|
Anandpara et al. [16] |
Case report |
India |
1 |
45 |
F |
Gradual loss of vision & proptosis |
Left |
3.7 |
Lateral orbitotomy |
Unspecified |
Albendazole |
Recovered |
10 |
|
|
Awad et al. [17] |
Case Series |
Egypt |
5 |
44 |
F |
Proptosis & diminished visual acuity |
Right |
N/A |
Transconjuctival incision |
Endocystectomy |
Topical antibiotics, steroid eye drops & NSAIDs |
Recovered |
58 |
|
|
13 |
M |
Proptosis, pain & diminished visual acuity |
Left |
N/A |
Transconjuctival incision |
Endocystectomy |
Topical antibiotics, steroid eye drops & NSAIDs |
Recovered |
42 |
|||||
|
11 |
M |
Proptosis & diminished visual acuity |
Left |
N/A |
Transconjuctival incision |
Endocystectomy |
Topical antibiotics, steroid eye drops & NSAIDs |
Recovered |
31 |
|||||
|
41 |
M |
Proptosis & diminished visual acuity |
Left |
N/A |
Transconjuctival incision |
Endocystectomy |
Topical antibiotics, steroid eye drops & NSAIDs |
Recovered |
23 |
|||||
|
39 |
F |
Proptosis, pain & diminished visual acuity |
Left |
N/A |
Transconjuctival incision |
Endocystectomy |
Topical antibiotics, steroid eye drops & NSAIDs |
Recovered |
11 |
|||||
|
|
|
|
|
18 |
F |
Proptosis |
Left |
N/A |
Lateral orbitotomy |
Unspecified |
Albendazole |
Recovered |
** |
|
|
62 |
F |
Proptosis |
Left |
N/A |
Lateral orbitotomy |
Unspecified |
Albendazole |
Recovered |
** |
|||||
|
33 |
F |
Proptosis |
Right |
N/A |
Lateral orbitotomy |
Unspecified |
Albendazole |
Recovered |
** |
|||||
|
44 |
F |
Proptosis |
Left |
N/A |
Inferior orbitotomy |
Unspecified |
Albendazole |
Recovered |
** |
|||||
|
26 |
M |
Proptosis |
Left |
N/A |
Lateral orbitotomy |
Unspecified |
Albendazole |
Recovered |
** |
|||||
|
Rajabi et al [30] |
Case report |
Iran |
1 |
23 |
F |
Severe proptosis |
Right |
N/A |
Lateral orbitotomy |
Total resection |
Albendazole |
Recovered |
48 |
|
|
Turgut et al. [31] |
Case report |
Turkey |
1 |
5 |
M |
proptosis |
Left |
N/A |
Transcranial approach |
Cystectomy with saline irrigation |
Mebendazole |
Had recurrence, recovered after conservative approach |
36 |
|
|
Arora et al. [32] |
Case report |
India |
1 |
16 |
M |
Impaired vision & dull headache |
Left |
N/A |
Curette evacuation |
Unspecified |
None |
Recovered |
N/A |
|
|
Lenztzsch et al. [33] |
Case report |
Germany |
1 |
5 |
F |
Proptosis, downward displacement of the eye |
Left |
N/A |
Lateral transosseous orbitotomy |
Unspecified |
Albendazole |
Recovered |
N/A |
|
|
Al-muala et al. [34] |
Case report |
Iraq |
1 |
42 |
F |
Swelling, proptosis, visual impairment & headache |
Right |
3 |
Lateral rhinotomy |
Cystectomy |
Albendazole |
Recovered |
8 |
|
|
Ahluwallaet al. [35] |
Case report |
India |
1 |
30 |
F |
Proptosis & headache |
Right |
2.5 |
Anterior orbitotomy with lateral extension |
Unspecified |
None |
Recovered |
N/A |
|
|
Sihota et al. [36] |
Case report |
India |
1 |
14 |
M |
Recurrent proptosis |
Left |
N/A |
No surgery was performed |
N/A |
Albendazole |
Had recurrence & recovered |
24 |
|
|
Huilgol et al. [37] |
Case report |
India |
1 |
8 |
F |
Proptosis, pain & diminished vision |
Right |
N/A |
Exenteration of the orbit |
N/A |
None |
Recovered |
N/A |
|
|
N/A: Not applicable, M: Male, F: Female, cm: Centimeter *Khan et al. gives a range of follow up periods between 3 to 12 months without specifying the exact periods of each patient. **Rajabi et al. gives a range of follow up periods between 2 to 6 years without specifying the exact periods of each patient. |
||||||||||||||
|
Variables |
Frequency (%)/mean ± SD |
|
Mean age |
27.45 ± 19.57 |
|
Age Group (years) 0-9 10-19 20-29 30-39 40-49 50-59 60-69 80-89 |
Number of patients (56) 11 (19.6%) 14 (25%) 9 (16.1%) 5 (8.9%) 8 (14.3%) 4 (7.2%) 4 (7.2%) 1 (1.8%) |
|
Gender Male Female |
Number of patients (56) 27 (48.2%) 29 (51.8%) |
|
Country of patients Afghanistan India Azerbaijan Morocco Turkey Egypt Iran Pakistan Syria Yemen Greece Ethiopia Germany Iraq |
Number of patients (56) 10 (17.9%) 8 (14.3%) 8 (14.3%) 6 (10.7%) 6 (10.7%) 5 (8.9%) 3 (5.36%) 3 (5.36%) 2 (3.57%) 1 (1.8%) 1 (1.8%) 1 (1.8%) 1 (1.8%) 1 (1.8%) |
|
Affected side Right side Left side |
Number of patients (56) 33 (58.9%) 23 (41.1%) |
|
Area of residency Urban Rural N/A |
1 (1.8%) 16 (28.6%) 39 (69.6%) |
|
Contact with sheep and dogs Reported N/A |
13 (23.2%) 43 (76.8%) |
Proptosis was present in 55 cases (98.2%), while visual impairment was reported in 37 cases (64.3%). Magnetic resonance imaging (MRI) was used for diagnosis in 38 cases (67.8%), while computed tomography (CT) was used in 28 cases (50%). Laboratory tests were conducted in 37 cases (66.1%), with 32 (86.5%) yielding normal results. The primary treatment for orbital HC was surgical removal of the cyst combined with anthelmintic therapy in 41 cases (73.2%). Surgery alone was performed in 14 cases (25%), while a conservative approach was used in one case (1.8%). Among those who underwent surgery, orbitotomy was the preferred surgical approach for accessing the cyst in 41 cases (74.5%). Cystectomy was the most common removal method, performed in 20 cases (36.4%), while the PAIR method (puncture, aspiration, injection, and re-aspiration) was used in 2 cases (3.6%). Follow-up durations ranged from 3 weeks to 72 months. Concurrent HC was reported in 2 cases (3.6%), while recurrence followed by recovery occurred in 4 cases (7.1%) (Table 3).
|
Variables |
Frequency (%) |
|
Presentation Symptomatic Asymptomatic |
Number of patients (56) 56 (100 %) 0 |
|
Common symptoms Proptosis Visual impairment |
Symptomatic patients (56) 55 (98.2%) 36 (64.3%) |
|
Imaging modalities MRI CT scan |
38 (67.8%) 28 (50%) |
|
Laboratory tests Positive Negative |
Number of patients (37) 5 (13.5%) 32 (86.5%) |
|
Mean cyst size (cm) ± SD |
3.25 ± 0.9 |
|
Therapeutic approach Surgery & anthelmintic drugs Surgery alone Conservative approach |
Number of patients (56) 41 (73.2%) 14 (25%) 1 (1.8%) |
|
Surgical technique for accessing the orbit Orbitotomy Trans-conjuctival incision Unspecified Combined approach Lateral rhinotomy Exenteration of the orbit Curette evacuation Transcranial approach |
Number of patients (55)
41 (74.5%) 5 (9.1%) 3 (5.5%) 2 (3.6%) 1 (1.8%) 1 (1.8%) 1 (1.8%) 1 (1.8%) |
|
Surgical technique for cyst removal Cystectomy Unspecified PAIR method Lynch method Aspiration and excision Barrett’s technique Total resection Aspiration and excision |
Number of patients (55) 20 (36.4%) 28 (50%) 2 (3.6%) 1 (1.8%) 1 (1.8%) 1 (1.8%) 1 (1.8%) 1 (1.8%) |
|
Anthelmintic drug of choice Albendazole Mebendazole |
Number of patients (42) 39 (92.9%) 3 (7.1%) |
|
Outcome Recovery N/A |
Number of patients (56) 55 (98.2%) 1 (1.8%) |
Discussion
Hydatid disease is a parasitic infection endemic in many regions worldwide. While traditionally attributed to Echinococcus granulosus, recent studies have identified five causative Echinococcus species with ten distinct genotypes (G1–G10), including E. oligarthrus, E. equinus, E. granulosus sensu stricto, E. canadensis, and E. felidis [4]. Orbital HCs are typically primary and occur unilaterally [7]. In endemic regions, HCs are the second most common cystic orbital lesions (25.8%), following dermoid cysts (29.7%) [8,9].
The clinical manifestations of HC primarily result from their mass effect on surrounding structures, especially in confined areas like the orbit. The predominant clinical manifestation of intra-orbital hydatid cysts, as observed in the present review, is a gradually progressive, unilateral proptosis, which may present in either an axial or non-axial orientation. This condition is generally painless, irreducible, non-pulsatile, and lacks blowing characteristics. If the cyst ruptures, it can cause inflammation. Additional symptoms of orbital HCs may include ocular pain, diplopia, headache on the affected side, blurred vision, vision loss, chemosis, eyelid edema, restriction of extraocular movements, and orbital cellulitis. In more advanced stages, signs may include optic disc swelling, optic atrophy with abnormal papillary defects, retinal vein engorgement, orbital bone erosion, hypopyon, and further eyelid edema [10]. The findings of the current review indicate that there is no evident sex predilection, as both males and females are affected at comparable rates. This observation aligns with existing literature; for instance, Khan et al. reported a case series in which 45.45% of the patients were female [9]. Although some suggest that the left side may be more prone to involvement due to the path of the left carotid artery [10], the findings of the current review indicate that the path of the left carotid artery does not predict which side will be affected, and there hasn’t been any definitive factor that can determine which side will be involved.
Children and young adults are the most commonly affected age groups; however, the condition is not limited to them. In the present review, the age of affected individuals ranged from three to 80 years, demonstrating the wide age distribution of the disease. Younger individuals may be more exposed to environments or activities that increase their risk of ingesting Echinococcus eggs, such as direct contact with infected animals (particularly dogs) or consumption of contaminated food or water. Additionally, they may be exposed to these risk factors for a longer duration, allowing sufficient time for HCs to form and grow before the disease develops. Cysts grow at an average rate of about 1–1.5 cm per year. Currently, there is no definitive categorization of “giant” HCs in the literature. Due to the limited space in the orbital cavity, patients typically develop symptoms within two years [10]. Orbital HCs are often diagnosed early in children due to the limited space within the orbit. The diagnosis of orbital HCs requires a combination of approaches, including laboratory tests, imaging, and histopathology for confirmation. Although various serological tests are available for the diagnosis of echinococcosis, their sensitivity is often limited in cases of orbital hydatid cysts. This limitation is evident in the present
review, where only five out of 37 serological tests produced positive results. They also have lower sensitivity compared to tests for other organs, as the parasitic proteins are less exposed to the immune system in the orbit [11].
Imaging tests, particularly MRI and CT, are the most commonly used modalities for diagnosing orbital HCs, a trend observed in the current review. On CT imaging, the lesion appears hypodense, unilocular, well-defined, and thin-walled, with a homogeneous mass featuring a hyperdense rim and capsular enhancement. On orbital MRI, the cyst demonstrates low signal intensity on T1-weighted images and high signal intensity on T2-weighted images, with contrast enhancement of the capsule [12]. MRI is superior to other imaging modalities as it provides more detailed information and can differentiate the cyst from other lesions and surrounding tissue. The differential diagnosis should include other cystic mass lesions, such as abscesses, mucoceles, intra-orbital hematomas, lacrimal tumors or cysts, and lymphangiomas [13].
Regarding treatment, surgical removal of the cyst without rupture is preferred. However, this is not always feasible due to the anatomical complexity of the orbit. The complex structure and thin walls of orbital HCs make them prone to rupture. Rupture may also result in the persistence of residual cyst wall fragments or cause secondary implantation of the parasite [14]. The PAIR method has emerged as a minimally invasive alternative for treating intra-abdominal HCs. However, for orbital HCs, as demonstrated in cases by Bamashmus et al. and Berradi et al., the PAIR method has been used out of necessity, primarily due to the anatomical constraints of the surgical area and the accidental rupture of the cyst [18,20]. Based on the results of the current review, orbitotomy is the preferred surgical approach for accessing and exploring the cyst in the orbit. However, various other techniques can be employed, with the choice of approach largely determined by the cyst's location, size, and the surgeon's expertise. Elkrimi et al. utilized a combined endoscopic and supraorbital incision approach to access a 3.1 cm cyst [22], while Mathad et al. and Al-Muala et al. accessed a 3 cm cyst using lateral orbitotomy and lateral rhinotomy, respectively [12,34]. The findings of the current review suggest that cystectomy is the preferred surgical technique for cyst removal. However, complications during the procedure can necessitate alterations in the surgical approach, requiring immediate modifications, as reported by Sendul et al [11].
Preoperative anthelmintic therapy, particularly with albendazole, is crucial for preventing parasite spread and reducing the risk of anaphylactic reactions in case of cyst rupture during surgery [12]. Postoperative administration of albendazole or mebendazole is also recommended to reduce the likelihood of relapse. Albendazole is commonly preferred due to its superior systemic absorption and better ability to penetrate cysts [10]. In the current review, Albendazole was used in 92.9% of the cases. Additionally, postoperative therapy included the use of steroids, NSAIDs, and antibiotics to manage symptoms, as shown by Awad et al. [17].
Regarding recurrence, the findings of this review suggest a higher likelihood of recurrence in cases with concurrent hydatidosis. The increased parasitic burden in these cases may be a significant contributing factor to disease recurrence. Preventing recurrence can be achieved by improving basic hygiene practices, such as handwashing after contact with dogs and sheep, enhancing livestock slaughter hygiene, ensuring continuous deworming of dogs, and promoting public education. During the course of this review, several limitations were identified. Firstly, most of the included papers, as well as the majority of the available literature, are case reports and case series. Additionally, a large amount of data was unretrievable during the search process.
Conclusion
Orbital HC is a rare condition, primarily diagnosed using MRI, with surgery as the definitive treatment. Concurrent hydatidosis increases the risk of recurrence, requiring thorough and ongoing follow-up.
Declarations
Conflicts of interest: The author(s) have no conflicts of interest to disclose.
Ethical approval: Not applicable, as systematic reviews do not require ethical approval.
Patient consent (participation and publication): Not applicable.
Funding: The present study received no financial support.
Acknowledgments: None to be declared.
Authors' contributions: EP, SHA, and OIM significantly contributed to the study's conception and the literature search for related studies. MMA, HAN, REM, and YMM were involved in the literature review, manuscript writing, and data analysis and interpretation. HA, SR, AA, and BA were involved in the literature review, the study's design, and the manuscript's critical revision. HAN and MMA confirm the authenticity of all the raw data. All authors approved the final version of the manuscript.
Use of AI: AI was not used in the drafting of the manuscript, the production of graphical elements, or the collection and analysis of data.
Data availability statement: Not applicable
References
- Nasralla HA, Abdalla BA, Abdullah HO, Ahmed SM, Kakamad FH, Mohammed SH, et al. Current Perspectives on Cystic Echinococcosis: A Systematic Review. Judi Clin. J. 2025;1(1):1-15. doi:10.70955/JCJ.2025.1
- Rawat S, Kumar R, Raja J, Singh RS, Thingnam SKS. Pulmonary hydatid cyst: Review of literature. J Fam Med Prim care. 2019 ;8(9):2774–8. doi:10.4103/jfmpc.jfmpc_624_19
- Abouassi M, Aloulou M, Hawa N, Toutounji T, Alyousef S. Successful eradication of a large orbital hydatid cyst without rupture using fronto-orbitozygomatic approach, the first case reported from Syria. Journal of Surgical Case Reports. 2020 ;2020(10):rjaa357. doi:10.1093/jscr/rjaa357
- Brunetti E, Kern P, Vuitton DA. Expert consensus for the diagnosis and treatment of cystic and alveolar echinococcosis in humans. Acta tropica. 2010;114(1):1-6. doi:10.1016/j.actatropica.2009.11.001
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Int J Surg. 2021 Apr; 88:105906. doi:10.1136/bmj.n71
- Kakamad FH, Abdalla BA, Abdullah HO, Omar SS, Mohammed SH, Ahmed SM, et al. Lists of predatory journals and publishers: a review for future refinement. European Science Editing. 2024;50:e118119. doi:10.3897/ese.2024.e118119
- Ilhami O, Jaoudy H, Razem B, Oukerroum A, Slimani F. Primary Intraorbital Hydatid Cyst: Case Reports. Medi Clin Case Rep J. 2024 ;2(2):318-20. doi:10.51219/MCCRJ/O-Ilhami/86
- Alabdullah MN, Awad A, Alabdullah H. Total Resection of Intraorbital Hydatid Cyst by Using Lynch Approach: A Case Report. Ear, Nose & Throat Journal. 2022 :01455613221113802. doi:10.1177/01455613221113802
- Khan T, Hussain I, Shah Z. Orbital Hydatid Cyst: An Interventional Case Series: Pakistan Journal of Ophthalmology. 2021 ;37(2). doi:10.36351/pjo.v37i2.1147
- Haydar AA, Rafizadeh SM, Rahmanikhah E, Nozarian Z, Aghajani A, Rajabi MT. Orbital intramuscular hydatid cyst causing compressive optic neuropathy: a case report and literature review. BMC ophthalmology. 2024 ;24(1):257. doi:10.1186/s12886-024-03502-w
- Sendul SY, Ucgul C, Dirim B, Demir M, Acar Z, Guven D. A difficult surgical approach for primary orbital hydatid cyst: transconjunctival medial orbitotomy. Journal of Surgical Case Reports. 2015 ;2015(12):rjv150.doi:10.1093/jscr/rjv150
- Mathad V, Singh H, Singh D, Butte M, Kaushik M. Large primary intraorbital hydatid cyst in elderly. Asian Journal of Neurosurgery. 2013 ;8(03):163-. doi:10.4103/1793-5482.121691
- Öztekin PS, Yilmaz BK, Gokharman FD, Koşar PN. Primary orbital hydatid cyst: computed tomography and magnetic resonance imaging findings. Singapore medical journal. 2014 ;55(11):e184. doi:10.11622/smedj.2014167
- Kumar M, Harsh V, Prakash A, Sahay CB, Kumar A. Neglected Case of Primary Intraorbital Hydatid Cyst. Neurology India. 2022 ;70(1):337-9. doi:10.4103/0028-3886.338694
- Debela AS, Abore KW, Worke AB, Wendimagegn ST. Primary Intra-orbital Hydatid cyst: a case report of a rare cause of Exophthalmos. International Medical Case Reports Journal. 2024 :89-92. doi:10.2147/IMCRJ.S454518
- Anandpara KM, Aswani Y, Hira P, Sathe PA. Isolated primary orbital hydatid disease presenting as multiple cystic lesions: a rare cause of proptosis. Annals of parasitology. 2015;61(3). doi:10.17420/ap6103.07
- Awad AA, Mohammad AE. A simple transconjunctival technique for the management of intraconal orbital hydatid cyst. Indian Journal of Ophthalmology. 2024:10-4103. doi:10.4103/IJO.IJO_756_24
- Bamashmus MA, Al-Shabbooti AA. Orbital hydatid cyst: an unusual presentation. Saudi Journal of Ophthalmology. 2006 ;20(2):146. doi:N/A
- Assimakopoulos SF, Marangos M. Orbital hydatid cyst. New England Journal of Medicine. 2020 ;382(14):1352-. doi:10.1056/NEJMicm1911903
- Berradi S, Hafidi Z, Lezrek O, Lezrek M, Daoudi R. Orbital hydatid cyst. QJM: An International Journal of Medicine. 2015 ;108(4):343-4. doi:10.1093/qjmed/hcu203
- Chtira K, Benantar L, Aitlhaj H, Abdourafiq H, Elallouchi Y. The surgery of intra-orbital hydatid cyst: a case report and literature review. Pan African Medical Journal. 2019 ;33(1). doi:10.11604/pamj.2019.33.167.18277
- Elkrimi Z, Douimi L, Labib O. Primary Intra-Orbital Hydatid Cyst as A Cause of Unilateral Exophthalmos: A Case Report. Medi Clin Case Rep J. 2024;2(2):210-2. doi:10.51219/MCCRJ/Elkrimi-Z/58
- Hosaini SF, Qader AQ, Neyazi M, Razaqi N, Satapathy P, Afzali H et al. Orbital hydatid cyst and its successful treatment: A case report from Afghanistan. American Journal of Ophthalmology Case Reports. 2024 ;36:102140. doi:10.1016/j.ajoc.2024.102140
- Jaffar N, Sattar S, Rahat N, Razzak S, Akram S. Rare Case of Orbital Echinococcosis. A Histopathological Perspective. Annals of Jinnah Sindh Medical University. 2018;4(2):89-91. doi: N/A
- Kars Z, Kansu T, Özcan OE, Erbengi A. Orbital echinococcosis: Report of two cases studied by computerized tomography. Journal of Neuro-Ophthalmology. 1982 ;2(3):197-200. doi: N/A
- Das D, Bhattacharjee K, Mohapatra SS, Islam S, Singh S, Deka A. Fluorescein staining in orbital echinococcosis. Indian Journal of Ophthalmology-Case Reports. 2022 ;2(4):1008. doi:10.4103/ijo.IJO_899_22
- Motlagh MF, Aghdam HJ, Motlagh BF. Primary orbital hydatid cyst: a case report. Acta Medica Iranica. 2017:530-2. doi: N/A
- Özek MM, Pamir MN, Sav A. Spontaneous rupture of an intraorbital hydatid cyst. A rare case of acute visual loss. J Clin Neuro-ophthalmol. 1993;13:135-7. doi: N/A
- Rajabi MT, Bazvand F, Makateb A, Hosseini S, Tabatabaie SZ, Rajabi MB. Orbital hydatid cyst with diverse locality in the orbit and review of literatures. Archives of Iranian Medicine. 2014 ;17(3):0-. doi: N/A
- Rajabi MT, Mohammadi S, Hassanpoor N, Hosseini SS, Shahbazi N, Tabatabaie SZ et al. Orbital alveolar echinococcosis: A case report. Journal of Current Ophthalmology. 2018 ;30(2):182-5. doi:10.1016/j.joco.2017.09.005
- Turgut M, Saglam S, Özcan OE. Orbital hydatid cyst: report of a case followed by serial computed tomography. Journal of Neuro-Ophthalmology. 1992 ;12(1):43-6. doi: N/A
- Arora MM, Dhanda RP, Bhagwat AG, Kalevar VK. Intra-ocular hydatid cyst. The British Journal of Ophthalmology. 1964 ;48(9):507. doi:10.1136/bjo.48.9.507
- Lentzsch AM, Göbel H, Heindl LM. Primary orbital hydatid cyst. Ophthalmology. 2016 ;123(7):1410. doi:10.1016/j.ophtha.2016.02.042
- Al-Muala HD, Sami SM, Shukri MA, Hasson HK, Alaboudy AT. Orbital hydatid cyst. Annals of maxillofacial surgery. 2012 ;2(2):197-9. doi:10.4103/2231-0746.101365
- Ahluwalla PP, Agrawal RV, Padmawar BU. Orbital hydatid cyst. Indian Journal of Ophthalmology. 1968 ;16(2):101-2. doi: N/A
- Sihota R, Sharma T. Albendazole therapy for a recurrent orbital hydatid cyst. Indian Journal of Ophthalmology. 2000 ;48(2):142-3. doi: N/A
- Huilgol AV. Orbital hydatid cyst. Indian Journal of Ophthalmology. 1963 ;11(3):79-81. doi: N/A

This work is licensed under a Creative Commons Attribution 4.0 International License.

