Primary Thyroid Non-Hodgkin B-Cell Lymphoma: A Case Series
Abstract
Introduction
Non-Hodgkin lymphoma (NHL) of the thyroid, a rare malignancy linked to autoimmune disorders, is poorly understood in terms of its pathogenesis and treatment outcomes. This study aims to review a single-center experience in managing primary thyroid non-Hodgkin B-cell lymphoma cases.
Methods
This retrospective case series was conducted at a single center from January 2020 to November 2024, including patients diagnosed with B-cell NHL of the thyroid who underwent surgical intervention. Data, including clinical, demographic, laboratory, and imaging information, were extracted from medical records. Diagnostic procedures involved core needle or surgical biopsy with immunohistochemistry analysis. Treatment included excisional biopsy, thyroidectomy, lobectomy, and chemotherapy. Quantitative data is presented as means and standard deviations, and qualitative data as frequencies and percentages.
Results
Among nine NHL cases, seven (77.8%) were female, with a mean age of 60.78 ± 12.53 years. Anterior neck swelling was the most common presentation in 6(66.7%) cases. Seven patients (77.8%) received R-CHOP chemotherapy; histopathology confirmed B-cell lymphoma in eight cases (88.9%). Thyroid function was euthyroid in four cases (44.4%), hypothyroid in three (33.3%), and hyperthyroid in one (11.1%). TI-RADS (Thyroid Imaging Reporting and Data Systems) classification showed five cases (55.6%) as TI-RADS 5. Follow-up revealed no recurrence in four cases (44.4%), and two deaths (22.2%).
Conclusion
Primary thyroid NHL is a rare condition requiring early diagnosis and personalized treatment. The variability in treatment responses highlights the need for individualized approaches to optimize patient outcomes.
Introduction
Non-Hodgkin lymphomas (NHLs) represent a heterogeneous group of lymphoid malignancies from lymphocytes, predominantly of B-cell origin. While many NHLs develop within lymph nodes, approximately 30-40% originate in extra-nodal sites [1]. Primary thyroid lymphoma is defined as a lymphoma that arises from the thyroid gland, excluding those that invade the thyroid due to either metastasis or direct extension. This rare malignancy accounts for approximately 5% of all thyroid malignancies and only 1-2% of all extra-nodal lymphomas, with an estimated annual incidence of 2 cases per million population [2].
The precise pathogenesis of primary thyroid lymphoma remains unclear. However, associations between autoimmune diseases, chronic antigenic stimulation, and the development of primary thyroid lymphoma have been identified. The most significant risk factor for primary thyroid lymphoma is the presence of hashimoto's thyroiditis, with patients affected having a risk of developing primary thyroid lymphoma that is 40 to 80 times higher. Notably, the incidence of hashimoto's thyroiditis among primary thyroid lymphoma patients is nearly 80% [3,4]. The proposed pathogenesis involves chronic antigenic stimulation from autoimmune processes leading to persistent lymphoid proliferation, eventually undergoing malignant transformation. Recent research has also implicated mutations in regulatory pathways, particularly the NF-κB signaling pathway through A20 gene mutations or deletions, in the development of certain thyroid B-cell lymphomas [5]
Primary thyroid lymphomas are almost exclusively NHL, with B-cell phenotype representing over 95% of cases [6]. Histologically, diffuse large B-cell lymphoma is the most common subtype, accounting for approximately 60-70% of cases. Meanwhile, mucosa-associated lymphoid tissue lymphoma represents the second most common subtype, comprising about 20-30% of cases. Other less common variants include Burkitt lymphoma [7]. These subtypes exhibit varying clinical behaviors and prognoses, necessitating accurate diagnosis for appropriate management.
Despite advances in understanding the pathophysiology and management of primary thyroid lymphoma, several knowledge gaps persist regarding optimal therapeutic strategies and long-term outcomes. This study aims to review a single-center experience in managing primary thyroid non-Hodgkin B-cell lymphoma cases. The eligibility of the references has been verified [8].
Methods
Study design and setting
This retrospective case series study was conducted at Smart Health Tower. It included patients diagnosed with NHL of B-cell origin in the thyroid gland based on pathological diagnosis who underwent surgical intervention, excisional biopsy, or thyroidectomy for definitive diagnosis. The study spanned from January 2020 to November 2024, with an average follow-up time of one year.
Data Collection
Data collection was performed over one month. Medical information, including demographic, clinical, and laboratory data, was retrospectively extracted from electronic medical records. Variables collected included past medic al history, surgical history, clinical presentation, thyroid status, thyroid function tests, autoimmune markers, fine needle aspiration findings, imaging studies, and treatment modalities. Pathological data were independently reviewed by an expert pathologist using specimen slides.
Laboratory and Imaging Assessments
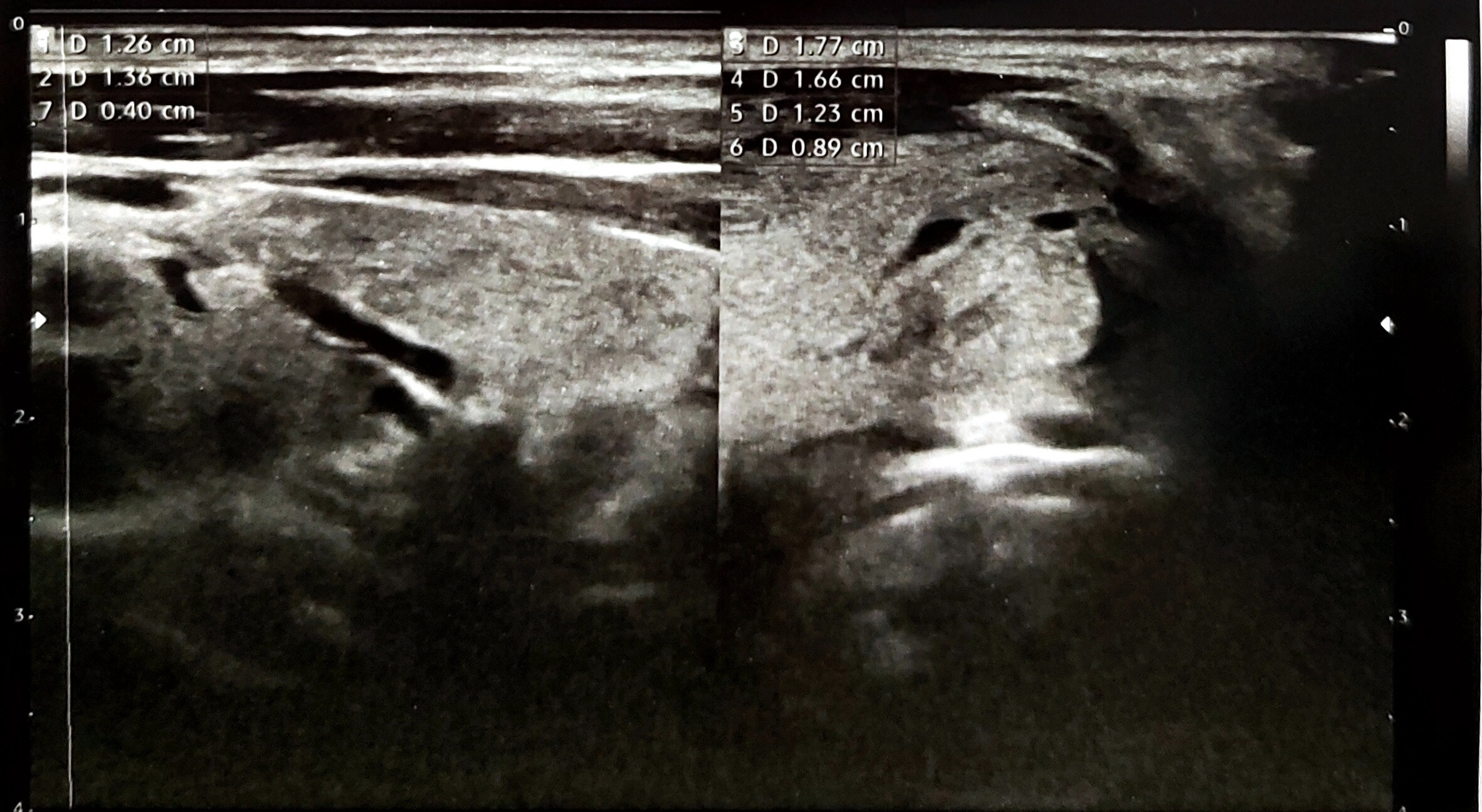
Comprehensive laboratory evaluations were conducted to assess thyroid function, autoimmune markers, and general health status. Thyroid function tests included thyroid-stimulating hormone and free thyroxine. Autoimmune markers measured included thyroid peroxidase antibodies and thyroglobulin antibodies. Additional tests included viral screenings and hemoglobin levels (complete blood count). Imaging studies included preoperative ultrasound to evaluate thyroid morphology, with hypoechoic lesions defined as having reduced echogenicity relative to normal thyroid tissue, while very hypoechoic lesions exhibited even lower echogenicity compared to adjacent musculature (Figure 1). Computed tomography (CT) and positron emission tomography (PET) scans were performed to assess disease extent in selected cases.
Diagnostic Biopsy Procedures
A core needle biopsy was performed using a spring-loaded Tru-Cut biopsy needle (18G). All patients underwent core needle or surgical biopsy for accurate diagnosis and immunohistochemistry analysis for markers.
Treatment Modalities
Surgical intervention primarily served a diagnostic role rather than a curative approach, with excisional biopsy, thyroidectomy, or lobectomy performed for tissue diagnosis. Chemotherapy protocols included the CHOP regimen, consisting of cyclophosphamide (750 mg/m²), adriamycin (50 mg/m²), vincristine (1.4 mg/m²), and prednisolone (100 mg/day), often combined with rituximab (375 mg/m²) to enhance therapeutic efficacy.
Ethical Considerations
The study was approved by the ethical committee of the Kscien organization. The study adhered to the principles of the Declaration of Helsinki. Due to its retrospective nature, neither patient approval nor informed consent was required.
Statistical analysis
The data were analyzed using the Statistical Package for the Social Sciences (SPSS) software version 27.0. Quantitative data were presented as means and standard deviations. Qualitative data were expressed as frequency and percentages.
Results
Among the nine cases, seven (77.8%) were female and two (22.2%) were male, with male-female ratio was 1:3.5. The mean age of patients was 60.78 ± 12.53 years. Clinically, anterior neck swelling was the most common presentation, observed in six cases (66.7%), while one case (11.1%) was on follow-up for preexisting thyroid conditions. The CT imaging revealed locally invasive thyroid masses involving the trachea, retrosternal space, or esophagus in three cases (33.3%), three cases (33.3%) had no available CT data. A history of prior thyroid surgery was noted in only one case (11.1%) (Table 1).
|
Cases |
Age (year) |
Gender |
PMH |
PSH |
Presentation |
Thyroid status |
Nodule location (Ultrasound) |
TI-RADS score |
CT scan |
PET scan |
|
Case 1 |
70 |
Female |
Hypothyroidism, Hypertension |
Thyroid surgery |
Follow Up |
Hypothyroid |
Right nodule |
TI-RADS 5 |
Right thyroid mass with tracheal invasion |
No abnormal metabolic activity, except in the thyroid gland |
|
Case 2 |
67 |
Male |
Type 2 diabetes mellitus, Colon cancer |
None |
Anterior neck swelling |
Euthyroid |
Bilateral nodule |
TI-RADS 5 |
Right thyroid mass with retrosternal extension, Suspicious left thyroid lesion |
No abnormal metabolically active lesion |
|
Case 3 |
79 |
Female |
Hypothyroidism, Atherosclerosis |
None |
Anterior neck swelling |
Hypothyroid |
Right nodule |
TI-RADS 5 |
Right thyroid mass with esophagus invasion |
No abnormal metabolically active lesion |
|
Case 4 |
62 |
Female |
Heart failure |
None |
Anterior neck swelling |
Euthyroid |
Bilateral nodule |
TI-RADS 3 |
Left lung focal ground glass appearance |
No abnormal metabolically active lesion |
|
Case 5 |
52 |
Female |
Hypertension |
None |
Anterior neck swelling |
Euthyroid |
Left nodule |
TI-RADS 4 |
NA |
No abnormal metabolically active lesion |
|
Case 6 |
47 |
Female |
Cerebrovascular accident |
None |
Anterior neck swelling |
Hyperthyroid |
Left nodule |
TI-RADS 5 |
Right thyroid mass |
NA |
|
Case 7 |
41 |
Female |
Systemic lupus erythematosus |
None |
Thyroid problem |
Hypothyroid |
Left nodule |
TI-RADS 2 |
NA |
No abnormal metabolically active lesion |
|
Case 8 |
72 |
Female |
Hydatid cyst |
None |
Thyroid problem |
Euthyroid |
Left nodule |
TI-RADS 5 |
Left thyroid nodule |
No abnormal metabolically active lesion |
|
Case 9 |
57 |
Male |
Negative |
None |
Anterior neck swelling |
NA |
Right nodule |
TI-RADS 4 |
NA |
No abnormal metabolically active lesion |
|
PMH: Past medical history, PSH: Past surgical history, NA: Not applicable, TI-RADS: Thyroid Imaging Reporting and Data Systems, CT: Computed tomography, PET: positron emission tomography |
||||||||||
Seven (77.8%) cases received R-CHOP chemotherapy, with six (66.7%) completing six or more sessions. Total thyroidectomy was performed in one case (11.1%). Histopathological examination confirmed B-cell lymphoma in eight cases (88.9%) and marginal zone lymphoma in one case (11.1%). Thyroid function testing revealed TSH abnormalities in four cases (44.4%), with one (11.1%) presenting significantly elevated TSH levels (100.0 mIU/L) (Table 2).
|
Cases |
TSH (mIU/L) |
Free T4 (ng/dL) |
TPO Ab (IU/mL) |
TG (ng/mL) |
CATN (pg/mL) |
Surgical management
|
Chemotherapy |
Histopathological examination type |
Histopathological examination sub-type |
Follow-up |
|
Case 1 |
7.01 |
11.7 |
NA |
169 |
0.5 |
NA |
R-CHOP 6 session |
NHL |
B-cell lymphoma |
No Recurrence |
|
Case 2 |
NA |
NA |
NA |
NA |
0.5 |
NA |
R-CHOP 6 session |
NHL |
B-cell lymphoma |
No Recurrence |
|
Case 3 |
100 |
0.97 |
372.6 |
0.04 |
0.5 |
NA |
R-CHOP 5 session |
NHL |
B-cell lymphoma |
Dead |
|
Case 4 |
0.57 |
15.2 |
NA |
NA |
NA |
NA |
R-CHOP |
NHL |
B-cell lymphoma |
NA |
|
Case 5 |
2.93 |
NA |
499 |
134 |
0.6 |
NA |
R-CHOP 6 session |
NHL |
B-cell lymphoma |
No Recurrence |
|
Case 6 |
0.039 |
18.54 |
NA |
NA |
NA |
NA |
Not take chemotherapy |
NHL |
B-cell lymphoma |
Dead |
|
Case 7 |
21.8 |
9.66 |
600 |
NA |
NA |
Total thyroidectomy |
R-CHOP 8 session |
NHL |
B-cell lymphoma |
No Recurrence |
|
Case 8 |
2.81 |
15.5 |
52.7 |
NA |
0.9 |
NA |
R-CHOP 9 session |
NHL |
B-cell lymphoma |
NA |
|
Case 9 |
NA |
NA |
NA |
NA |
NA |
NA |
5 biology + 15 radiotherapy |
NHL |
Marginal zone lymphoma |
NA |
|
TSH: Thyroid stimulating hormone, TPO Ab: Thyroid Peroxidase Antibodies, TG: Thyroglobulin, CATN: Calcitonin, NHL: Non-Hodgkin Lymphoma, R-CHOP: Rituximab- Cyclophosphamide Doxorubicin Vincristine Prednisone, NA: Not Available |
||||||||||
Thyroid function status was euthyroid in four cases (44.4%), hypothyroid in three cases (33.3%), and hyperthyroid in one case (11.1%). Nodule location based on ultrasound was left-sided in four cases (44.4%), right-sided in three cases (33.3%), and bilateral in two cases (22.2%). The TI-RADS (Thyroid Imaging Reporting and Data Systems) classification showed that five cases (55.6%) were TI-RADS 5, while two (22.2%) were TI-RADS 4. The mean Free T4 and TSH levels were 11.93 ± 6.02 ng/dL and 19.31 ± 36.36 mIU/L, respectively. Regarding follow-up outcomes, four cases (44.4%) showed no recurrence, and two (22.2%) resulted in mortality (Table 3).
|
Variables |
Frequency (%) |
|
Age (Mean ± SD) |
60.78 ± 12.53 |
|
Gender Male Female |
7 (77.8) |
|
Thyroid status Euthyroid Hypothyroid Hyperthyroid NA |
4 (44.4) 3 (33.3) 1 (11.1) 1 (11.1) |
|
TI-RADS score TI-RADS 2 TI-RADS 3 TI-RADS 4 TI-RADS 5 |
1 (11.1) 2 (22.2) 5 (55.6) |
|
Free T4 (Mean ± SD) |
11.93 ± 6.02 |
|
TSH level (Mean ± SD) |
19.31 ± 36.36 |
|
TPO Ab (Mean ± SD) |
381.08 ± 237.86 |
|
CATN (Mean ± SD) |
0.60 ± 0.17 |
|
Follow-up No Recurrence Dead NA |
4 (44.4) 2 (22.2) 3 (33.3) |
|
Nodule location (Ultrasound) Right nodule Left nodule Bilateral nodule |
3 (33.3) 4 (44.4) 2 (22.2) |
|
PET scan findings Thyroid gland lesion No abnormal active lesion NA |
1 (11.1) 7 (77.8) 1 (11.1) |
|
TI-RADS: Thyroid Imaging Reporting and Data System, TSH: Thyroid stimulating hormone, TPO: Thyroid Peroxidase, CATN: Calcitonin, SD: Standard deviation, NA: Not available |
|
Discussion
Primary thyroid lymphoma is confined to the thyroid gland, with or without local lymph node involvement in the neck. There is no evidence of distant metastasis at the time of initial diagnosis. Epidemiological studies have consistently shown a higher prevalence of primary thyroid lymphoma (including NHL) in females compared to males, with a female-to-male ratio ranging from 1.8 to 4.4:1, predominantly affecting individuals in their fifth to eighth decades of life [9,10]. In line with these findings, the present study also noted a female predominance, a female-to-male ratio of 3.5:1, and an average age of 60.78 years.
The primary risk factor for primary thyroid lymphoma, including NHL, is Hashimoto's thyroiditis, which significantly increases the likelihood of developing it by 40 to 80 times. Despite this elevated risk, only 0.6% of individuals with Hashimoto’s thyroiditis go on to develop primary thyroid lymphoma [11,12]. The condition is predominantly associated with hypothyroidism, especially in the context of Hashimoto's thyroiditis, whereas cases of primary thyroid lymphoma occurring in hyperthyroid or euthyroid states are rare. This association is thought to arise due to the thyroid’s lack of native lymphoid tissue, which may accumulate due to chronic antigenic stimulation in Hashimoto’s thyroiditis, leading to lymphoid infiltration and the subsequent risk of lymphoma [10]. In the present study, of the nine cases of NHL, 5(55.6%) were euthyroid or hyperthyroid, with four patients being euthyroid and one hypothyroid. This supports the notion that while hypothyroidism is commonly associated with NHL, other thyroid states, including euthyroid and hyperthyroid, can also occur in these cases.
Patients with thyroid NHL typically present with a rapidly enlarging cervical mass that remains mobile during swallowing. This thyroid enlargement may result in compressive symptoms, including dysphagia, dyspnea, and hoarseness [13,14]. The mass can sometimes exert pressure on venous structures, leading to facial puffiness or swelling. Furthermore, lymphoma infiltration of the thyroid can induce hypothyroidism, manifesting as fatigue, cold intolerance, and dry skin. Additionally, systemic symptoms commonly associated with lymphoma, such as fever, night sweats, unexplained weight loss, and generalized pruritus, may also be observed [15]. In the current study, anterior neck swelling was the most common presentation, observed in six cases (66.7%). The high prevalence of anterior neck swelling in this study likely reflects the tumor's location in the thyroid, causing significant local enlargement and compressive effects.
Diagnosis of NHL of the thyroid necessitates a multifaceted approach, integrating clinical findings, advanced imaging techniques, and definitive tissue sampling. Imaging modalities such as ultrasound and CT scans are crucial in identifying thyroid abnormalities and associated lymphadenopathy [16]. Diagnosis is typically confirmed through histological examination and immunohistochemical results [10]. Identifying immunoglobin clonal gene rearrangements is essential for the differential diagnosis in patients with Hashimoto thyroiditis and a histologically benign lymphoepithelial lesion. Core needle biopsy or surgical excision is required to obtain sufficient tissue for accurate diagnosis and subtyping [17]. The present study used a comprehensive diagnostic approach, including ultrasound, CT scans, positron emission tomography scans, and fine needle aspiration cytology to detect thyroid abnormalities and associated lymphadenopathy. Core needle biopsy was performed when fine needle aspiration cytology alone was insufficient for diagnosis. Histopathological and immunohistochemical evaluations confirmed the presence of lymphoma.
Management of NHL of the thyroid typically involves a combination of approaches, including chemotherapy, radiation therapy, and, in some cases, surgery. The choice of treatment depends on the stage and grade of the lymphoma and the patient's overall health. In current practice, surgery mainly serves the purpose of obtaining tissue for diagnosis [2]. Radiotherapy, being highly effective for local disease control, is often utilized due to the radiosensitive nature of thyroid NHL. In contrast, chemotherapy targets occult or systemic disease, thereby enhancing long-term outcomes. The conventional CHOP regimen, comprising Cyclophosphamide, Doxorubicin, Vincristine, and Prednisolone, remains the standard treatment approach for systemic disease management [15,18]. Following three to six cycles of chemotherapy, radiation therapy is commonly administered to enhance disease control. The introduction of Rituximab has shown promising efficacy, particularly in elderly patients with diffuse large B-cell lymphoma. However, the role of surgical intervention remains debated [19]. Several factors influence prognosis, including patient age, tumor grade, and disease stage. Notably, mucosa-associated lymphoid tissue lymphoma generally exhibits a more favorable prognosis than large B-cell lymphoma, and outcomes are typically better in pediatric and young adult patients [20].
Recent studies on outcomes of primary thyroid lymphoma underscore substantial prognostic heterogeneity influenced by histopathological classification, therapeutic approach, and clinical factors. A population-based study of 1,408 primary thyroid lymphoma (including NHL) patients reported a median survival of 9.3 years, with multivariate analysis identifying advanced age, disease stage, histological subtype, and treatment modality as independent prognostic determinants [3]. Diffuse large B-cell lymphoma, the most prevalent subtype comprising approximately 68% of cases, exhibits variable 5-year survival rates between 45% and 90%, contingent on treatment protocols [4]. Notably, combined-modality therapy, particularly rituximab-based immunochemotherapy with radiation, has significantly enhanced prognosis, achieving 5-year overall and progression-free survival rates of 81.2% and 77.8%, respectively, compared to monotherapy approaches. In contrast, indolent subtypes such as mucosa-associated lymphoid tissue lymphoma demonstrate intermediate 5-year survival rates of approximately 62%, though they remain susceptible to persistent recurrence, unlike aggressive variants, which exhibit cure probabilities exceeding 90% after three years of remission [9]. Molecular analyses have further elucidated distinct evolutionary pathways underlying thyroid lymphoma relapse, providing insight into the observed variations in treatment response and clinical outcomes [21]. These findings collectively support the development of risk-adapted treatment algorithms, highlighting the necessity of histology-directed, multimodal therapeutic strategies to optimize survival while mitigating treatment-associated morbidity in this rare lymphoproliferative malignancy [22].
This retrospective case series of primary thyroid B-cell NHL is subject to several limitations. The short follow-up period of one year restricts the ability to evaluate long-term outcomes, recurrence patterns, and survival rates. Additionally, as a single-center study, the findings may lack broader applicability across different healthcare settings. The heterogeneity in treatment modalities, coupled with the absence of molecular and genetic analyses, further limits insights into the pathophysiology of the disease and optimal therapeutic strategies.
Conclusion
This study underscores the significance of early diagnosis and tailored treatment for primary thyroid NHL. The findings highlight the variable thyroid function and treatment responses, underscoring the need for personalized approaches to optimize patient outcomes.
Declarations
Conflicts of interest: The author(s) have no conflicts of interest to disclose.
Ethical approval: The study was approved by the ethical committee of the Kscien organization (No.32).
Patient consent (participation and publication): Written informed consent was obtained from patients for publication.
Source of Funding: Smart Health Tower.
Role of Funder: The funder remained independent, refraining from involvement in data collection, analysis, or result formulation, ensuring unbiased research free from external influence.
Acknowledgements: None to be declared.
Authors' contributions: AMS and SFA were significant contributors to the conception of the study and the literature search for related studies. RMA, HOB, IJH, DOK, ROM and SHH were involved in the literature review, the study's design, the critical revision of the manuscript, they participated in data collection. AMM, HAA, and AAQ were involved in the literature review, study design, and manuscript writing. AJQ was the radiologists who performed the assessment of the cases. AMA was the pathologist who performed the histopathological diagnosis. AMS and SHH confirm the authenticity of all the raw data. All authors approved the final version of the manuscript.
Use of AI: AI was not used in the drafting of the manuscript, the production of graphical elements, or the collection and analysis of data.
Data availability statement: Not applicable
References
- Shankland KR, Armitage JO, Hancock BW. Non-hodgkin lymphoma. The Lancet. 2012;380(9844):848-57. doi:10.1016/s0140-6736(12)60605-9
- Lee JS, Shin SJ, Yun HJ, Kim SM, Chang H, Lee YS, Chang HS. Primary thyroid lymphoma: A single-center experience. Frontiers in endocrinology. 2023; 14:1064050. doi:10.3389/fendo.2023.1064050
- Graff-Baker A, Roman SA, Thomas DC, Udelsman R, Sosa JA. Prognosis of primary thyroid lymphoma: demographic, clinical, and pathologic predictors of survival in 1,408 cases. Surgery. 2009;146(6):1105-15. doi:10.1016/j.surg.2009.09.020
- Walsh S, Lowery AJ, Evoy D, McDermott EW, Prichard RS. Thyroid lymphoma: recent advances in diagnosis and optimal management strategies. The oncologist. 2013;18(9):994-1003. doi:10.1634/theoncologist.2013-0036
- Kuribayashi-Hamada Y, Ishibashi M, Tatsuguchi A, Asayama T, Takada-Okuyama N, Onodera-Kondo A, et al. Clinicopathologic characteristics and A20 mutation in primary thyroid lymphoma. Journal of Nippon Medical School. 2022;89(3):301-8. doi:10.1272/jnms.jnms.2022_89-305
- Pedersen RK, Pedersen NT. Primary non‐Hodgkin's lymphoma of the thyroid gland: a population- based study. Histopathology. 1996;28(1):25-32. doi:10.1046/j.13652559.1996.268311.x
- Cha H, Kim JW, Suh CO, Kim JS, Cheong JW, Lee J, et al. Patterns of care and treatment outcomes for primary thyroid lymphoma: a single institution study. Radiation oncology journal. 2013;31(4):177. doi:10.3857/roj.2013.31.4.177
- Kakamad FH, Abdalla BA, Abdullah HO, Omar SS, Mohammed SH, Ahmed SM, et al. Lists of predatory journals and publishers: a review for future refinement. European Science Editing. 2024;50:e118119. doi:10.3897/ese.2024.e118119
- Alzouebi M, Goepel JR, Horsman JM, Hancock BW. Primary thyroid lymphoma: the 40 year experience of a UK lymphoma treatment centre. International journal of oncology. 2012;40(6):2075-80. doi:10.3892/ijo.2012.1387
- Hirokawa M, Suzuki A, Hashimoto Y, Satoh S, Canberk S, Jhuang JY, et al. Prevalence and diagnostic challenges of thyroid lymphoma: a multi-institutional study in non-Western countries. Endocrine journal. 2020;67(11):1085-91. doi:10.1507/endocrj.ej20-0202
- Lanham T, Lanham E, Sullivan A, Magaji V. Non-Hodgkin lymphoma of the thyroid in a patient with hyperthyroidism. Journal of Community Hospital Internal Medicine Perspectives. 2021;11(1):79-80. doi:10.1080/20009666.2020.1829403
- Noureldine SI, Tufano RP. Association of Hashimoto's thyroiditis and thyroid cancer. Current opinion in oncology. 2015;27(1):21-5. doi:10.1097/cco.0000000000000150
- Rosai J, Ackerman LV, Goldblum JR, Lamps LW, McKenney JK, Myers JL. Rosai and Ackerman’s Surgical Pathology. Philadelphia: Elsevier; 2018
- Derringer GA, Thompson LD, Frommelt RA, Bijwaard KE, Heffess CS, Abbondanzo SL. Malignant lymphoma of the thyroid gland: a clinicopathologic study of 108 cases. The American journal of surgical pathology. 2000;24(5):623-39. doi:10.1097/00000478-200005000-00001
- Katna R, Shet T, Sengar M, Menon H, Laskar S, Prabhash K, D'Cruz A, Nair R. Clinicopathologic study and outcome analysis of thyroid lymphomas: experience from a tertiary cancer center. Head & neck. 2013;35(2):165-71. doi:10.1002/hed.22928
- Mahmoud EM, Howard E, Ahsan H, Cousins JP, Nada A. Cross-sectional imaging evaluation of atypical and uncommon extra-nodal head and neck non-Hodgkin lymphoma: Case series. Journal of Clinical Imaging Science. 2023; 13:6. doi:10.25259/JCIS_134_2022
- Prasad BK, Ranganathan Y, Dwivedi G, Mukherjee T, Pathak A. Primary Thyroid Lymphoma: An Interesting Case Emphasising Early Diagnosis. Bengal Journal of Otolaryngology and Head Neck Surgery. 2022;30(3):345-8. doi:10.47210/bjohns.2022.v30i3.821
- Peixoto R, Pinto JC, Soares V, Koch P, Gomes AT. Primary thyroid lymphoma: a case report and review of the literature. Annals of Medicine and Surgery. 2017; 13:29-33. doi:10.1016/j.amsu.2016.12.023
- Chen C, Tibbetts KM, Tassler AB, Schiff BA. Tracheal invasion and perforation from advanced primary thyroid lymphoma: a case report and literature review. American Journal of Otolaryngology. 2013;34(5):559-62. doi:10.1016/j.amjoto.2013.04.004
- Hedhili F, Kedous S, Jbali S, Attia Z, Dhambri S, Touati S, et al. Primary thyroid lymphoma: case report and review of the literature. Egyptian Journal of Ear, Nose, Throat and Allied Sciences. 2015;16(2):171-3. doi:10.1016/j.ejenta.2015.05.002
- Tzioni MM, Watanabe N, Chen Z, Wu F, Madej E, Makker J, et al. Primary thyroid B‐cell lymphoma: molecular insights into its clonal evolution and relapse. The Journal of Pathology. 2025;265(2):123-31. doi:10.1002/path.6380
- Kumar R, Khosla D, Kumar N, Ghoshal S, Bera A, Das A, et al. Survival and failure outcomes in primary thyroid lymphomas: a single centre experience of combined modality approach. Journal of thyroid research. 2013;2013(1):269034. doi:10.1155/2013/269034

This work is licensed under a Creative Commons Attribution 4.0 International License.

