Arteriovenous Fistula Creation for Hemodialysis in Patients with End-Stage Renal Disease with and Without Surgical Drain: A Randomized Control Trial
Abstract
Introduction
Failure of an arteriovenous fistula (AVF) disrupts hemodialysis access and reduces the available area for future access. Preventive interventions are necessary to avoid AVF failure. This study evaluates the impact of surgical drainage during AVF creation for hemodialysis in patients with end-stage renal disease (ESRD).
Methods
This single-center, phase II, randomized controlled trial was conducted from June 2020 to June 2023. Ninety-four patients were randomly assigned into two groups: Group A (with a surgical drain) and Group B (without a drain). Patients were followed for six months post-surgery. The primary outcome was AVF primary patency, and secondary outcomes included postoperative complications.
Results
The average age of participants was 63.7 years, with 50 male patients. The most common cause of renal failure was glomerular disease (29.8%), and most AVFs were located on the left side (57.4%). Brachiocephalic AVFs were the most frequent type (70.2%). Postoperative hematoma was more common in Group B (42.6%) than in Group A (17%) (P = 0.007). The primary patency rate at six months was higher in Group A (87.2%) compared to Group B (76.6%), though the difference was not statistically significant (P = 0.180).
Conclusion
The use of surgical drainage during AVF creation may reduce postoperative complications, such as hematomas, and potentially improve primary patency rates, contributing to better outcomes for patients undergoing hemodialysis.
Introduction
Renal failure is a serious public health problem, and its incidence is increasing. Nowadays, hemodialysis (HD) and kidney transplantation are the main therapies for end-stage renal disease (ESRD) [1]. Regardless of the rise in kidney transplant surgeries, HD remains the mainstay of treatment. In the majority of cases, a phase of hemodialysis preceded the transplantation [2]. Patients who depend on HD require proper vascular access. According to the National Kidney Foundation-Dialysis Outcomes Quality Initiative (NKF-DOQI) recommendations, optimal vascular access should offer an appropriate flow rate, durability, and a low risk of complications [3].
There are three main types of chronic vascular access for HD, including native arteriovenous fistula (AVF), arteriovenous shunts employing graft material (AV graft), and central venous catheter (CVC). Among them, AVF stands out as the primary vascular access worldwide, given its superior long-term primary patency rate, minimal need for secondary procedures, and its association with longer survival rates and lower complication rates [4,5]. The NKF-DOQI recommended the radiocephalic fistula in the nondominant forearm as the primary choice for access [6]. With the growing emphasis on AVF and the evolving dialysis population, which now includes a higher proportion of older patients with cardiovascular comorbidities, upper arm fistulas have gained popularity in recent years [7]. The cephalic vein is superficial in the forearm and is easily injured by previous venipunctures, making the creation of radiocephalic AVF difficult. Hence, with the ability to protect the cephalic vein in the arm, a brachiocephalic AVF becomes a practical alternative procedure. [8]. Currently, brachiocephalic AVF is increasing in popularity because of the higher failure rate of radiocephalic fistulas [9]. Insufficient vascular access and associated consequences have been identified as the cause of mortality in about 25% of all patients initiating HD. [5]. Failure of an AVF not only disrupts functional access but also reduces the available area from which another access may be established. Furthermore, interventional techniques must be performed on the patients to repair the failure of AVFs. As a result, minimizing post-operative complications that impact AVF patency and failure rates is of critical importance [9].
The current study aims to assess the overall outcomes and effects of surgical drainage in AVFs for hemodialysis patients with renal failure.
Methods
Study design and setting
This was a single-center, phase II, open-label, parallel-arm, randomized controlled trial (RCT) conducted between June 1, 2020, and June 1, 2023, involving ESRD patients in need of AVF for hemodialysis or maintenance hemodialysis. The trial aimed to investigate the outcomes and complication rates of AVFs with and without postoperative surgical drains in patients undergoing AVF creation. The study was conducted in accordance with the Helsinki Declaration. The study proposal was approved by the scientific and ethical committee of B.P. Koirala Institute of Health Sciences. All patients consented to participate in the trial and the publication of their information. The study's details have been registered in the Chinese Clinical Trial.
Participants
All patients who underwent native AVF creation were included in this study. Participants were excluded if they matched any of the following criteria: (1) having a bleeding disorder; (2) history of antiplatelet use; (3) previous AVF creation; (4) cephalic vein diameter less than 3 mm.
Randomization and masking
Once eligibility was established, the patient's electronic file was initially admitted to a designated mailbox in hospital's database. The second registration was completed after confirming all preoperative requirements for inclusion by computerized assignment. The participants were assigned randomly (1:1) into two groups, Group A (inserting a surgical drain at the site of the AVF) or Group B (without a surgical drain). The final registration was done when the patient was discharged home, followed up regularly, and met all the inclusion criteria. No masking of the operators or participants in the allocation was performed.
Preoperative assessment
All patients underwent clinical examination to assess the adequacy of the venous and arterial systems of the upper limbs. If the vein was not visible, duplex scanning was requested. Basic investigations, including a complete blood count, viral markers, and electrolytes, were done for all of the cases. Preoperative antibiotic (Cefepime 1gm iv) was given to all of the patients.
Procedure
In the supine position, under local anesthesia (15 cc lidocaine 2%) using a transverse antecubital incision, an end-to-side AVF was created. One cc of heparin (5000 IU) was injected before arterial clamping, and no reversal agent was used in the completion of the procedure. Six zero Prolene with an 8 mm needle was used as the suture material in all procedures. A Redivac drain (size 18 in brachiocephalic and size 16 in radiocephalic AVF) was inserted in the subcutaneous tissues inferolateral to the incision. The patient remained in the hospital overnight. Postoperatively, they were given oral analgesics and antibiotics for five days in accordance with the hospital infection prevention protocol.
Outcome
Postoperatively, patients were followed up regularly for six months. The primary outcome was the primary patency of the AVF, while the secondary outcomes included postoperative complications such as hematoma, pain, reopening, and wound infection.
Statistical analysis
The database of the hospital was used to collect patient data. The collected data were analyzed using the Statistical Package for the Social Sciences 25.0 software. The qualitative data were presented in the form of frequency and percentages, and the Chi-square (X2) test was used to compare them. A P-value of less than 0.05 is considered significant.
Results
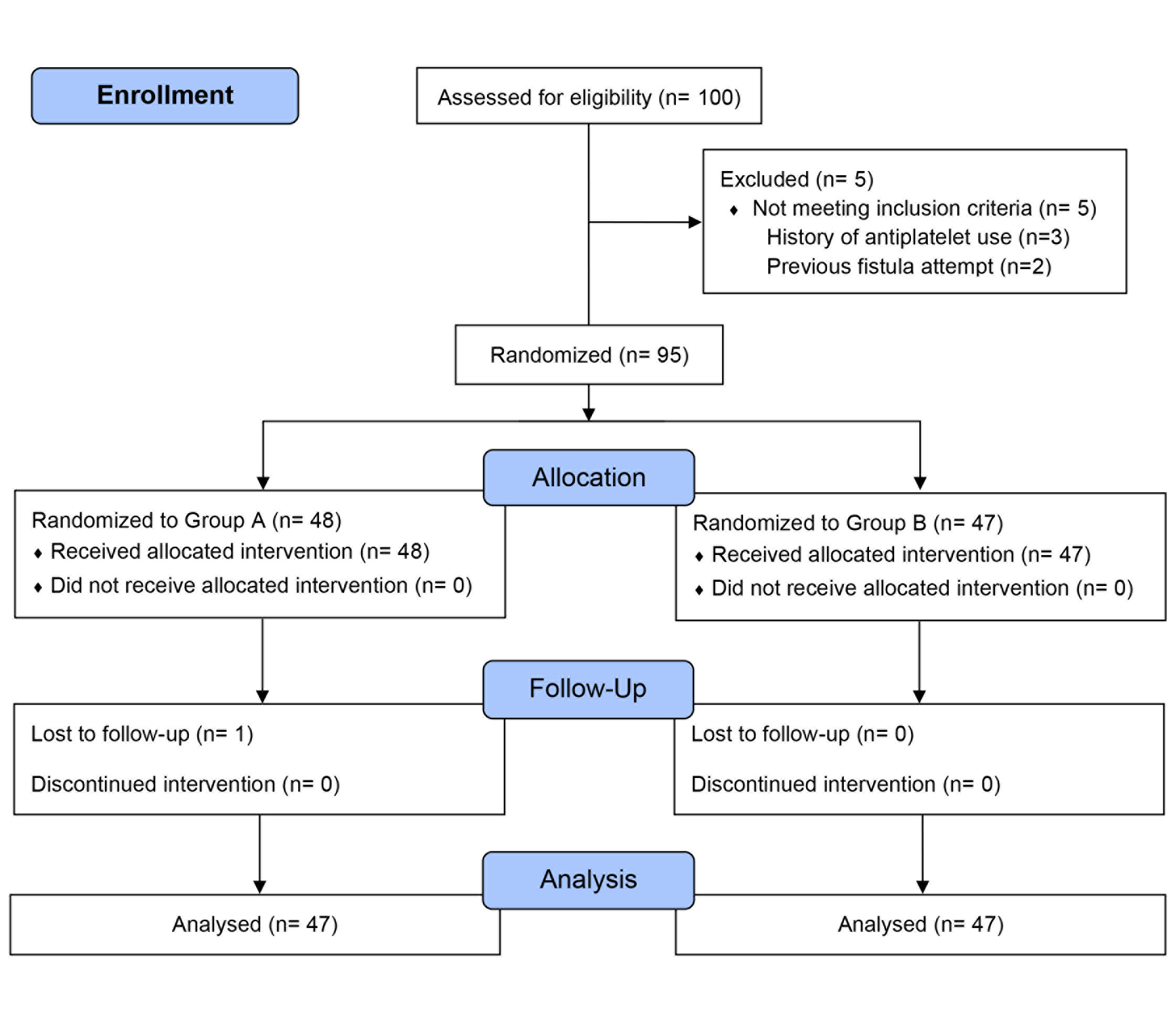
During the follow-up period, 94 patients were registered for the trial. The mean age of the patients was 63.7 years ranging from 44 to 81 years. Fifty cases (53.2%) were male and 44 (46.8%) were female. The most common cause of renal failure was glomerular disease (29.8%), followed by diabetic nephropathy (22.3%), and analgesic nephropathy (11.7%). Fifty-four (57.4%) patients had a history of temporary vascular access (CV line) (Table 1). The majority of the AVFs were located on the left side (57.4%). Brachiocephalic was the most common type of AVF (70.2%) followed by radiocephalic fistula (18.1%) (Table 2). Postoperative hematoma was more common in Group B (42.6%) than in Group A (17%) and reached a significant level (P-value = 0.007). About 10.6% of cases in Group B underwent reopening of the fistula while none of the cases of Group A underwent reopening. Although the difference wasn’t statistically significant, the primary patency rate at six months was relatively higher in Group A (87.2%) than in Group B (76.6%) (P-value 0.180) (Table 3). One patient developed an infection which was in the experimental group. In the experimental group, individuals experiencing primary failure were somewhat older, with ages ranging from 55 to 74 years, compared to the total participants. Five of the six cases of primary failure in the experimental group were female (83.3%).
|
Variables |
Group A |
Group A |
P-value |
|
Age, years, mean ± SD |
62.6 ± 8.99 |
64.9 ± 9.25 |
0.954 |
|
Sex Male Female |
24 (51.1%) 23 (48.9%) |
26 (55.3%) 21 (44.7%) |
0.679 |
|
On hemodialysis Yes No |
25 (53.2%) 22 (46.8%) |
29 (61.7%) 18 (38.3%) |
0.404 |
|
Primary renal disease Glomerular Interstitial Analgesic nephropathy Diabetic nephropathy ADPKD Vascular Others |
15 (31.9%) 3 (6.4%) 6 (12.8%) 8 (17%) 4 (8.5%) 5 (10.6%) 6 (12.8%) |
13 (27.7%) 2 (4.3%) 5 (10.6%) 13 (27.7%) 3 (6.4%) 4 (8.5%) 7 (14.9%) |
0.924 |
|
Variables |
Group A |
Group A |
P-value |
|
Site of AV fistula Right site Left site |
18 (38.3%) 29 (61.7%) |
22 (46.8%) 25 (53.2%) |
0.404 |
|
Type of fistula BC RC RB |
33 (70.2%) 10 (21.3%) 4 (8.5%) |
33 (70.2%) 7 (14.9%) 7 (14.9%) |
0.510
|
|
Variables |
Group A |
Group A |
P-value |
|
Hematoma Yes |
8 (17%) 39 (83%) |
20 (42.6%) 27 (67.4%) |
0.007 |
|
Reopening Yes No |
0 (0%) 47 (100%) |
5 (10.6%) 42 (89.4%) |
0.022 |
|
Pain Mild Moderate Severe |
38 (80.9%) 8 (17%) 1 (2%) |
36 (76.6%) 8 (17%) 3 (6.4%) |
0.590 |
|
Primary patency Yes No |
41 (87.2%) 6 (12.8%) |
36 (76.6%) 11 (23.4%) |
0.180 |
Discussion
Around the world, there is a continuous increase in the number of ESRD patients admitted for renal replacement therapy. Because HD is the recommended treatment for the great majority of these patients, permanent vascular access is the only means to survive. As a result, the effective creation of permanent functional vascular access is essential for providing adequate HD therapy in ESRD [10]. A well-functioning AVF is ideal vascular access for HD and has a major influence on patient outcome and survival [11]. Patients’ survival and quality of life are also impacted by vascular access complications. Therefore, the appropriate management to decrease the complications is mandatory [12]. However, as the life expectancy of patients undergoing HD has increased over time, many of them will require additional vascular access operations throughout their lives [6]. The distal radiocephalic AVF is the preferred vascular access, followed by other alternative accesses. However, multiple factors, including obesity, unavailability, exhaustion, and calcified vessels make alternative vascular access mandatory [13]. The primary issue with AVF has always been the high risk for early thrombosis, which results in early failure [1]. Other common consequences influencing AVF patency include stenosis, thrombosis, bleeding, infection, and flow problems [13].
Ates et al. discovered that the brachiocephalic group had higher complications than the radiocephalic group. However, for hematoma, the situation was reversed, as it occurred in 5.9% of the brachiocephalic group and 6.9% of the radiocephalic group without a significant effect [2]. Thabet et al. reported hematoma in 20 (8.4%) of their patients. Seventeen (85%) patients were effectively treated with hematoma evacuation and repair of the puncture site. Because of late presentation with possibly contaminated hematomas, the remaining three (15%) patients had their access ligated [14]. The bleeding rate in the studies by Magar et al., Elamurugan et al., and Madhhachi et al. were 9.75%, 11.5%, and 5.3% respectively [5,6,10]. In the current study, hematoma was more common in the experimental group than in the control group, and the result was statistically significant (P-value = 0.007).
A significant problem with AVFs is the high rate of primary failure, which can be caused by a lack of maturation or early thrombosis [15]. A comprehensive strategy is essential in detecting and addressing the principal causes of primary failure in individuals with ESRD. Despite current research outlining the pathophysiology of the technique and biomechanical challenges connected with maturation, the process of AVF maturation remains complicated and poorly understood. Intimal hyperplasia has been identified as the most severe pathohistological alteration that occurs in blood vessels and has been linked to AVF primary failure [9]. In a study that compared the primary patency of radiocephalic AVF and brachiocephalic AVF, brachiocephalic AVF had the highest patency rate (79.18%), followed by mid-arm radiocephalic AVF (72%), and distal arm radiocephalic AVF (68.18%) [5]. In a meta-analysis of 46 reports, the probability of primary failure was 23%, but it raised to 37% in old-aged patients [16,17]. According to Zouaghi et al., the actual primary patency rates at six months, 1 year, 2years, 4years, and 5years were 82%, 78%, 69%, 61%, and 42%, respectively [18]. Wong et al. reported that primary patency at three months and one year for brachiocephalic fistula was 87.9% and 63.1%, respectively, and 84.6% and 58.1% for radiocephalic fistula [19]. Mahalkar et al. reported brachiocephalic AVF patency of 88%, 83%, and 71% at 30 days, 90 days, and six months, respectively [20]. The current study's primary patency rate at six months was 87.2% for the control group, which was slightly higher than the control group's rate of 76.6%.
Age, gender, race, diabetes, peripheral vascular disease, history of coronary artery disease, location of the fistula, and obesity are all patient factors that predict primary failure [17]. However, some studies reported that age did not affect primary patency [14,21]. Smith et al.’s review of the literature revealed an increase in access failure in the elderly population [22]. There is limited evidence that AVF patency varies by gender. Several studies discovered that when the patency rate was examined by gender, male patients had a much greater rate than female patients [23]. This finding is explained by the fact that females have a smaller diameter of arteries and so have a lower AVF patency rate than males [22]. However, Mortaz et al. found no evidence that AVF survival was gender-dependent [24]. In the experimental group of the current study, those with primary failure were somewhat older (ages ranging from 55 to 74 years) than the total participants. Five of the six cases of primary failure in control group were female, which might indicate that females are more prone to failure than males.
Infection is responsible for 20% of all AVF consequences. The majority of AVF infections involve perivascular cellulitis, which often appears as localized erythema and edema and is easily treated. An infection linked to anatomical abnormalities such as aneurysms, hematomas, or abscesses is far more dangerous and necessitates surgical excision and drainage [25]. The infection rates in studies by Dekhaiya et al. and Schinstock et al. were 8% and 26.8%, respectively [26,27]. In a study by Shameri et al., infection was observed in 17 (7.4%) of the cases, with the majority of the cases (10, 4.4%) being managed with observation (antibiotic) or aspiration and draining. Other seven (3%) infections progressed to ruptured fistulae, which required emergent surgical treatment [9]. Another study reported that 57 (23.8%) patients had severe infections in the form of abscess formation or active bleeding. As a result, conservative therapy was out of the option, and they all had immediate access closure [14]. Only one patient developed an infection in the current study, which was in the experimental group. The patient was treated conservatively with antibiotics for five days and responded well to the treatment.
One of the present study's limitations was the sample size which was small and only covered participants from a single center. However, because this is a hypothesis-generating study, more study on this concept is needed.
Conclusion
The use of surgical drainage after AVF surgery might be beneficiary. It may decrease the complications associated with AVF creation and improve the fistula's primary patency.
Declarations
Conflicts of interest: The author(s) have no conflicts of interest to disclose.
Ethical approval: The study was approved by the B.P. Koirala Institute of Health Sciences Ethical Committee (No.11).
Patient consent (participation and publication): Written informed consent was obtained from patients for publication.
Source of Funding: B.P. Koirala Institute of Health Sciences.
Role of Funder: The funder remained independent, refraining from involvement in data collection, analysis, or result formulation, ensuring unbiased research free from external influence.
Acknowledgements: None to be declared.
Authors' contributions: LSJ was significant contributor to the conception of the study and the literature search for related studies. NO and SV were involved in the literature review, the study's design, the critical revision of the manuscript, they participated in data collection, involved in the literature review, study design, and manuscript writing. SV and LSJ confirm the authenticity of all the raw data. All authors approved the final version of the manuscript.
Use of AI: AI was not used in the drafting of the manuscript, the production of graphical elements, or the collection and analysis of data.
Data availability statement: Not applicable.
References
- Bashar K, Medani M, Bashar H, Ahmed K, Aherne T, Moloney T, et al. End-to-side versus side-to-side anastomosis in upper limb arteriovenous fistula for dialysis access: a systematic review and a meta-analysis. Annals of vascular surgery. 2018; 47:43-53. doi:10.1016/j.avsg.2017.07.036".2017.07.036
- Ates A, Özyazicioglu A, Yekeler I, Ceviz M, Erkut B, Karapolat S, et al. Primary and secondary patency rates and complications of upper extremity arteriovenous fistulae created for hemodialysis. The Tohoku journal of experimental medicine. 2006;210(2):91-7. doi:10.1620/tjem.210.91"10.1620/tjem.210.91
- Ahmed OF, Hamodat OM, Kakamad FH, Abduljabbar RS, Salih AM, Omar DA, et al. Outcomes of arteriovenous fistula for hemodialysis in pediatric age group. Annals of Medicine and Surgery. 2021;72:103100. doi:10.1016/j.amsu.2021.103100".2021.103100
- Khadatkar A, Mahakalkar C, Pradhan T, Bora A. Comparative analysis of radiocephalic versus brachiocephalic native arteriovenous fistula for hemodialysis in end stage renal disease. Int J Res Med Sci. 2016;5:171. doi:10.18203/2320-6012.ijrms20164543
- Al-Madhhachi BA. The outcome of radiocephalic after brachiocephalic and redo arteriovenous fistula. SAGE Open Medicine. 2022;10:20503121211069280. doi:10.1177/20503121211069280
- Elamurugan E, Hemachandar R. Brachiocephalic arteriovenous fistula through the median antecubital vein for hemodialysis. Indian Journal of Vascular and Endovascular Surgery. 2016;3(2):49. doi:10.4103/0972-0820.183645
- Hu D, Li C, Sun L, Zhou C, Li X, Ai Z, et al. A modified nontransposed brachiobasilic arteriovenous fistula versus brachiocephalic arteriovenous fistula for maintenance hemodialysis access. Journal of Vascular Surgery. 2016;64(4):1059-65. doi:10.1016/j.jvs.2016.03.450
- Sabah MN, Ahmed SP, Khan MS, Hasan R, Hossain MF, Fatma MN. A Comparative Study between Radiocephalic and Brachiocephalic Arteriovenous Fistula in End Stage Renal Disease. Cardiovascular Journal. 2021;14(1):44-9. doi:10.3329/cardio.v14i1.55373
- Al-Shameri I, KhudaBux G, Al-Ganadi A. Prospective Evaluation of Factors Associated with Arteriovenous Fistula Primary Failure and Complications in Hemodialysis Patients: A Single Center-Study. Cardiol Vasc Res. 2021;5(3):1-7. doi:N/A
- Magar DT, Shrestha K, Chapagain D, Shrestha K, Thapa S. Comparative Study of Autologous Radiocephalic and Brachiocephalic Arteriovenous Fistula in Patients with End Stage Renal Disease. Journal of Nepalgunj Medical College. 2020;18(1):78-81. doi:10.3126/jngmc.v18i1.35206
- Beathard GA, Litchfield T, Jennings WC. Two-year cumulative patency of endovascular arteriovenous fistula. The journal of vascular access. 2020;21(3):350-6. doi:10.1177/1129729819877780
- Ramanathan AK, Nader ND, Dryjski ML, Dosluoglu HH, Cherr GS, Curl GR, et al. A retrospective review of basilic and cephalic vein-based fistulas. Vascular. 2011;19(2):97-104. doi:10.1258/vasc.2010.oa0228
- Eldesouky MS, Fayed A. Comparative study between brachiocephalic fistula and proximal radiocephalic fistula for hemodialysis in patients with end-stage renal disease. The Egyptian Journal of Surgery. 2019;38(1):33-8. doi:10.4103/ejs.ejs_92_18
- Thabet BA, Ewas MO, Hassan HA, Kamel MN. Complications of arteriovenous fistula in dialysis patients at Assiut University Hospital. Journal of Current Medical Research and Practice. 2017;2(2):119. doi:10.4103/JCMRP.JCMRP_32_16
- Mahakalkar CC, Jajoo SN, Kaple MN, Kshirsagar S, Philip M, Thorat V. Brachiobasalic Versus Brachiocephalic Arteriovenous Fistula for Vascular Access during Hemodialysis: A Review. Indian Journal of Forensic Medicine & Toxicology. 2021;15(3). doi:10.37506/ijfmt.v15i3.15425
- Al-Jaishi AA, Oliver MJ, Thomas SM, Lok CE, Zhang JC, Garg AX, et al. Patency rates of the arteriovenous fistula for hemodialysis: a systematic review and meta-analysis. American Journal of Kidney Diseases. 2014;63(3):464-78. doi:10.1053/j.ajkd.2013.08.023".2013.08.023
- MacRae JM, Dipchand C, Oliver M, Moist L, Lok C, Clark E, et al. Arteriovenous access failure, stenosis, and thrombosis. Canadian journal of kidney health and disease. 2016; 3:2054358116669126. doi:10.1177/2054358116669126
- Zouaghi MK, Lammouchi MA, Hassan M, Rais L, Krid M, Smaoui W, et al. Determinants of patency of arteriovenous fistula in hemodialysis patients. Saudi Journal of Kidney Diseases and Transplantation. 2018;29(3):615. doi:10.4103/1319-2442.235183
- Wong FL, MacAulay E, Hussey K. TP5. 2.4 Patency of radiocephalic versus brachiocephalic arteriovenous fistula in a major tertiary hospital. British Journal of Surgery. 2021;108(7): 362-010. doi:10.1093/bjs/znab362.010
- Mahakalkar C, Jajoo SN, Kaple M, Kshirsagar S. Vascular access for dialysis-A choice between brachiobasilic versus brachiocephalic arteriovenous fistula. Journal of Datta Meghe Institute of Medical Sciences University. 2022;17(1):84. doi:10.4103/jdmimsu.jdmimsu_233_22
- Gibson KD, Gillen DL, Caps MT, Kohler TR, Sherrard DJ, Stehman-Breen CO. Vascular access survival and incidence of revisions: a comparison of prosthetic grafts, simple autogenous fistulas, and venous transposition fistulas from the United States Renal Data System Dialysis Morbidity and Mortality Study. Journal of vascular surgery. 2001;34(4):694-700. doi:10.1067/mva.2001.117890
- Smith GE, Gohil R, Chetter IC. Factors affecting the patency of arteriovenous fistulas for dialysis access. Journal of vascular surgery. 2012;55(3):849-55. doi:10.1016/j.jvs.2011.07.095".2011.07.095
- Tsuchida K, Nagai K, Yokota N, Okada D, Muromiya Y, Suenaga T, et al. Simple surgical method for a native arteriovenous fistula of chronic hemodialysis patients and the patency rate. The journal of vascular access. 2015;16(10):13-7. doi:10.5301/jva.5000408
- Mortaz SS, Davati A, Ahmadloo MK, Taheri HR, Golfam F, Tavakoli A, et al. Evaluation of patency of arteriovenous fistula and its relative complications in diabetic patients. Urology Journal. 2013;10(2):894-7. doi:N/A
- Aljuaid MM, Alzahrani NN, Alshehri AA, Alkhaldi LH, Alosaimi FS, Aljuaid NW, et al. Complications of arteriovenous fistula in dialysis patients: Incidence and risk factors in Taif city, KSA. Journal of Family Medicine and Primary Care. 2020;9(1):407. doi:10.4103/jfmpc.jfmpc_848_19
- Dekhaiya FA, Hathila TN, Doshi SA, Mehta NB, Shah SM, Rupani MP. A prospective study of arteriovenous fistula creation in chronic renal failure patients in Bhavnagar, Gujarat, western India. International Journal of Medical Science and Public Health. 2016;5(02):1. doi:10.5455/ijmsph.2016.1507201543
- Schinstock CA, Albright RC, Williams AW, Dillon JJ, Bergstralh EJ, Jenson BM, et al. Outcomes of arteriovenous fistula creation after the Fistula First Initiative. Clinical Journal of the American Society of Nephrology. 2011;6(8):1996-2002. doi:10.2215/CJN.11251210

This work is licensed under a Creative Commons Attribution 4.0 International License.
