Gastric Pyloric Schwannoma: A Case Report and Review of the Literature
Abstract
Introduction
Schwannomas are slow-growing, subclinical neoplasms rarely found in the gastrointestinal tract. This study reports a schwannoma in the pyloric region of the stomach.
Case presentation
A 50-year-old female presented with a one-week history of epigastric pain, dark tarry stools, and nausea. Endoscopic examination and biopsy confirmed the diagnosis of gastric schwannoma. The patient underwent surgical resection of the tumor. Histopathological examination showed benign spindle cells with strong S100 positivity, confirming schwannoma. Post-operative follow-up included treatment for H. pylori infection and monitoring for recurrence or complications. No recurrence was reported after six months.
Literature Review
Gastric schwannoma is challenging to distinguish from other submucosal tumors preoperatively. Reviews of recent case reports indicate the importance of detailed imaging in diagnosis, and surgical resection remains the treatment of choice, with an excellent prognosis and low recurrence rates.
Conclusion
Schwannoma is rare in the stomach, especially in the pyloric region. Definitive diagnosis may require immunohistochemical analysis. Appropriate follow-up after treatment can be essential to identify emerging complications and ensure timely intervention.
Introduction
Schwannomas, also referred to as neurilemmomas or neurinomas, are uncommon and non-cancerous spindle cell tumors that originate from excessive proliferation of myelin-producing Schwann cells in the nerve sheath and remain localized in their primary location [1,2]. They are frequently slow-growing and rarely found in the gastrointestinal tract (GIT). Gastric schwannoma (GS) represents only 2–6% of mesenchymal tumors within the GIT and 0.2% of all gastric tumors [3,4]. Although most schwannomas occur alone, GS is often part of neurofibromatosis type 2 and has an association with other tumors. There is a gender predilection towards females [3]. Preoperatively, GS is often challenging to accurately distinguish from gastric submucosal or other stromal tumors due to physicians' limited recognition of GS [5,6]. Herein, a case of schwannoma originating from the pyloric region of the stomach is reported. The references’ eligibility has been verified, and the report has been structured in accordance with CaReL guidelines [7,8].
Case Presentation
Patient information
A 50-year-old female presented with epigastric pain for one week. The pain was associated with melena and nausea, with no constipation, diarrhea, or fever. Her past medical history was negative for any chronic disease. She had a thyroid lobectomy two years ago and was on thyroxin 100 mcg/day.
Clinical findings
Only epigastric tenderness was noted on physical examination, with no other systemic abnormalities.
Diagnostic assessment
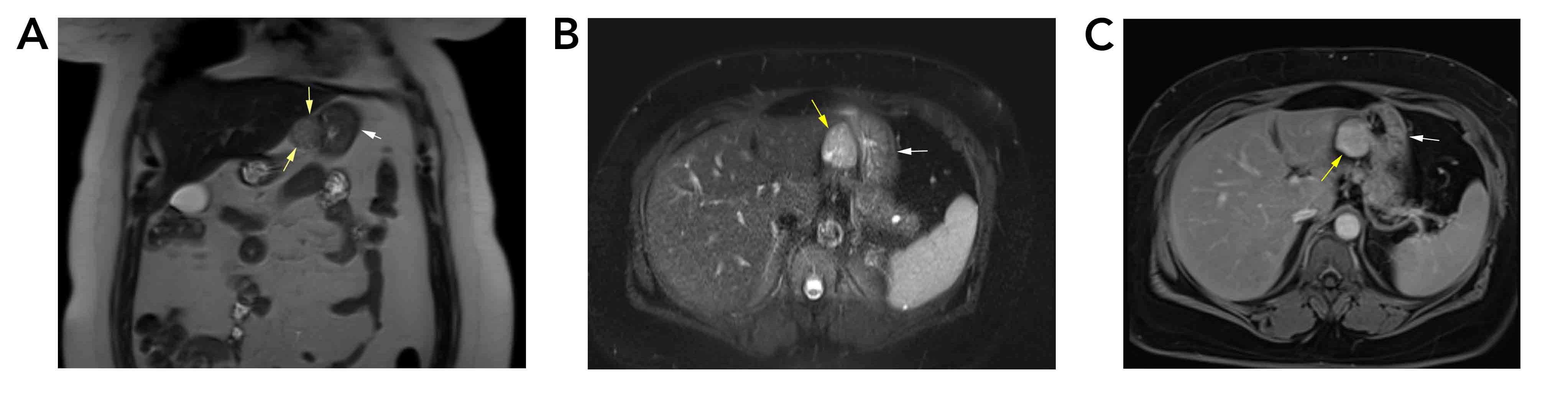
An abdominal ultrasound (U/S) showed a well-defined hypoechoic mass (30 mm) in the pyloric region of the stomach. A contrast-enhanced computed tomography (CT) scan of the abdomen revealed a well-defined, smooth outline lesion measuring 35x30 mm in the pyloric region with mild mucosal wall thickening (7 mm) and relative proximal dilation of the stomach, without lymphadenopathy. A dynamic magnetic resonance imaging (MRI) of the abdomen revealed a well-defined, 33 x 27 x 27 mm, space-occupying lesion in the epigastric region between the lesser curvature of the stomach and the left hepatic lobe. The mass exhibited T1 hypointensity, T2 hyperintensity, restricted diffusion on diffusion-weighted imaging, and diffuse early enhancement with retained contrast in the delayed phase. The mass was attached to the stomach wall (Figure 1). An esophagogastroduodenoscopy (EGD) revealed a large subepithelial lesion, approximately 4 cm, located in the incisura and extending to the lower body on the lesser curvature side, with an antral nipple sign. The overlying mucosa was normal, but there was nodular antral gastropathy.
Therapeutic intervention
A resection of the anterior gastric wall near the incisura angularis was performed to remove the mass. Histopathological examination of the lesion revealed hypo- and hypercellular areas of spindled cells arranged in loose fascicles and having neural-type, lightly eosinophilic and clear cytoplasm and spindled and buckled nuclei with fine chromatin. Scattered hyalinized blood vessels were present within the lesion. These findings indicated a benign spindle cell lesion suggestive of schwannoma. The tumor had a mitotic rate of less than five mitoses per 50 high-power fields. There was necrosis or vascular invasion, and the resection margins were free. Four lymph nodes were examined and found to be negative for metastasis (Figure 2). Immunohistochemistry of the tumor revealed positivity for S100, characterized by strong and diffuse cytoplasmic and nuclear staining. Weak and focal cytoplasmic staining was observed for desmin and smooth muscle actin, while the tumor was negative for CD117 and CD34.
![Figure 2. A) The tumor is composed of alternating hypercellular and hypocellular areas. B) The cells have a spindle shape and are arranged in loose fascicles. There is a hyalinized blood vessel. C-D) The cells have indistinct cytoplasmic membranes with lightly eosinophilic to clear cytoplasm. The nuclei are spindled, wavy, and buckled, with fine chromatin. [Hematoxylin and eosin; original magnification x 40 (A), x 100 (B), x 400 (C and D)].](https://www.barwmedical.com/public/site/images/12ayman12/mceu-63543796741743423581812.jpg)
Follow-up and Outcome
Six months postoperatively, the patient developed lower back pain. Lumbosacral MRI showed L3-4 and L4-5 mild disc thecal sac indentation, causing mild bilateral foraminal narrowing and a focal bone lesion at the acetabular root. Pelvic MRI indicated early osteoarthritis changes in the right hip joint with subchondral pseudocystic changes at the acetabular roof. It also showed a thin-walled unilocular cystic lesion in the left ovary measuring 40x20 mm. The patient received analgesics for disc problems and osteoarthritis. A follow-up EGD one year later revealed a long transverse scar in the incisura area and mucosal congestion. Multiple biopsies from the area showed chronic active H. pylori pangastritis without metaplastic and dysplastic changes. The patient was referred to a gastroenterologist and started a regimen for H. pylori eradication. Subsequently, a CT scan revealed a thickening of the gastric outlet and pyloric wall. The rest of the stomach was distended and fluid-filled, raising suspicion of a gastric ulcer. An endoscopic biopsy from the gastric mucosa revealed mild chronic gastritis with mild glandular atrophy, ulceration, and fibromuscular hyperplasia, with no evidence of H. pylori microorganisms, intestinal metaplasia, or dysplasia. A biopsy was also taken from a small mid-esophageal ulcer, which showed no pathologic findings.
Discussion
Gastric schwannomas are rare mesenchymal benign tumors, comprising 0.2% of all gastric tumors [4]. Malignant transformation of GS is extremely rare [9]. The development of GS can occur at any age, but it is most commonly found in individuals in their fifties and sixties, with a higher incidence in women. Although many GSs are discovered incidentally, they may cause nonspecific symptoms such as pain or GIT bleeding [6]. In reviewing eight cases of GS, four cases presented with abdominal pain or discomfort, two had a palpable abdominal mass, one had hematochezia, and another one had weight loss and early satiety (Table 1). The present case was a 50-year-old female who complained about abdominal pain in the epigastric region for a week.
|
Author, year [Reference] |
Study design |
No. of cases |
Age (year) |
Sex |
Clinical Presentation |
History |
Diagnostic Assessment |
Treatment |
Follow up-time |
Recurrence |
||||
|
Medical |
Surgical |
Mass Location/ Detection method |
Disease appearance |
Mass size (mm) |
Immunohistochemical findings |
|||||||||
|
Sorial et al., 2024[1] |
Case report |
1 |
82 |
M |
Abdominal pain |
N/A |
N/A |
GC of the stomach / CT |
No lesions or ulcers |
32 |
S100 (+). CD34, CD117, DOG-1, DS, & SMA (-) Ki67 (2-3%) |
SWGR during laparoscopy |
N/A |
N/A |
|
Majdoubi et al., 2024[3] |
Case report |
1 |
50 |
M |
Persistent postprandial pain, melena, anorexia, asthenia |
None |
None |
Fundic region along the GC/ CT, GI endoscopy |
Peptic ulcer and lesion |
33 |
Fusiform cells. CD 117 (-) S100 (+) DOG (-) DS (-) AML (-) |
Laparoscopic gastric atypical resection |
8 months |
No |
|
Manjesh et al., 2024[4] |
Case report |
1 |
62 |
F |
Early satiety, weight loss |
N/A |
N/A |
Fundus / USG, upper GI endoscopy, CECT |
Hypoechoic mass |
83 |
Spindle cells. S100, GFAP, & P16 (+). CD117, DOG-1, SMA, & CD34 (-)
|
Exploratory laparotomy & Excision |
N/A |
N/A |
|
Huang et al., 2024[5] |
Case report |
1 |
72 |
F |
Abdominal distension |
N/A |
N/A |
Anterior lower stomach near GC / EUS, CT, EG |
White ulcer scar |
55 |
S-100, SDHB, & SOX-10 (+) CD34, CD117, DOG-1, & DS (−). Ki67 (1%) |
EFTR |
1 week |
No |
|
Kostovski et al., 2024[6] |
Case report |
1 |
68 |
M |
Lower abdominal discomfort |
None |
None |
Antral region along the GC / CECT |
Submucosal gastric lesion |
62 |
Spindle cells. S100, SDHB, & SOX10, (+). CD34, CD 117, DOG-1, & DS (−). Ki-67 (<1%) |
Supraumbilical median laparotomy & Excision |
1 month |
No |
|
Kormann et al., 2024[10] |
Case report |
1 |
67 |
M |
Progressive fatigue, paleness, hematochezia |
None |
N/A |
Gastric antrum / Upper EUS |
Submucosal lesion |
9 |
S100 (+) |
Hybrid ESD-EFTR |
1st 6 years
|
No |
|
2nd 3 months after intervention |
||||||||||||||
|
Lin et al., 2024[11] |
Case report |
1 |
66 |
F |
Protruding subepithelial mass |
N/A |
N/A |
Gastric fundus / EGD, EUS, CT |
Subepithelial lesion |
25 |
Short spindle-shaped cells. S100 (+)
|
Laparoscopic surgery |
N/A |
N/A |
|
Huang et al., 2024[12] |
Case report |
1 |
31 |
F |
Palpable abdominal mass |
N/A |
N/A |
Posterior wall of the GLC/ USG, CT |
Mucosal ulcer |
64 |
N/A |
Laparoscopic gastric lesion resection |
2 weeks |
No |
|
M: male F: female IDA: iron-deficiency anemia N/A: non-available GC: greater curvature CT: computed tomography GLC: gastric lesser curvature USG: ultrasonography EUS: endoscopic ultrasound EGD: esophagogastroduodenoscopy GI: gastrointestinal CECT: contrast-enhanced computed tomography EG: electrocoagulation mm: millimeter (+): positive (-): negative GFAP: glial fibrillary acidic protein DS: desmin SMA: smooth muscle actin ESD: endoscopic submucosal dissection EFTR: Endoscopic full-thickness resection SWGR: stapled wedge gastric resection |
||||||||||||||
Diagnosing GSs presents several challenges due to their rare occurrence and nonspecific symptoms. Due to overlapping clinical, radiological, and endoscopic features, these tumors are often misidentified as other submucosal tumors, such as gastrointestinal stromal tumors, leiomyomas, or other mesenchymal tumors, which might bring delay in availing the right therapy. Endoscopic ultrasound is a valuable tool for differentiation but is not definitive. Schwannomas are typically diagnosed postoperatively through histopathological and immunohistochemical analysis, with S100 and SOX-10 providing positive confirmation and negative markers such as CD117 and smooth muscle actin, which help exclude other differential diagnoses [5,10]. Among the reviewed cases, CT was one of the most common methods used for the early characterization of the lesion. It was used in 87.5% of the cases, providing detailed imaging that helped identify the tumor's size, location, and relation to surrounding structures. Ultrasonography was used in 62.5% of cases, with 60% being endoscopic U/S, which aided in characterizing the lesion and determining its layer of origin, as this is essential for planning the therapeutic approach. Other methods, like EGD and upper gastrointestinal endoscopy, were less commonly used but played supportive roles in the diagnostic process [4,11]. In the current case, an abdominal U/S initially identified a well-defined hypoechoic mass in the pyloric region of the stomach. This was followed by a contrast-enhanced CT scan of the abdomen, which confirmed a smooth contoured lesion and mild mucosal wall thickening. Dynamic MRI showed a 33 x 27 x 27 mm lesion in the epigastric region between the lesser curvature of the stomach and the left hepatic lobe. An EGD revealed a large subepithelial lesion with an antral nipple sign.
Primarily, GS occurs in the fundus and body of the stomach, as documented in multiple case reports [1,3-5,11,12]. In three cases, including those by Majdoubi et al., Lin et al., and Manjesh et al., the tumor was located in the fundus [3,4,11], while in three other cases, the tumor was located in the gastric body [1,5,12].
The antrum can also be the site of tumor origin, as described in two cases by Kormann et al. and Kostovski et al. [6,10]. No cases were reported in the cardia or pylorus. However, in the present case, the tumor originated from the pyloric region.
In the current case, a surgical resection of the anterior gastric wall near the incisura angularis was performed, and a 4.2-cm tumor was successfully removed. Histopathological examination showed a neural-type, low-grade, spindle cell tumor with a low mitotic rate, no necrosis or vascular invasion, and with negative margins. Immunohistochemical analysis revealed strong S100 positivity, insignificant staining for desmin and SMA, and negativity for CD117 and CD34, confirming the diagnosis of schwannoma. This approach aligns with other studies that reported successful surgical resection outcomes and similar histological and immunohistochemical profiles [1,4-6].
Post-surgical monitoring was crucial in the current case. The need to address new health concerns, such as H. pylori eradication, aligns with the follow-up practices noted by Kormann et al., who emphasized the importance of routine imaging and endoscopy to monitor for recurrence and manage complications [10]. However, unlike the present case, Lin et al. reported no significant post-surgical complications or new health issues during follow-up, suggesting a variation in the clinical outcomes [11]. Although detailed imaging and histopathological analyses were conducted in the current case, genetic profiling of the tumor was not performed. This could have provided valuable information about the tumor’s potential for malignant behavior and recurrence.
Conclusion
Schwannoma is rare in the stomach, especially in the pyloric region. Definitive diagnosis may require immunohistochemical analysis. Appropriate follow-up after treatment can be essential to identify emerging complications and ensure timely intervention.
Declarations
Conflicts of interest: The author(s) have no conflicts of interest to disclose.
Ethical approval: Not applicable.
Patient consent (participation and publication): Written informed consent was obtained from the parent of the patient for publication.
Funding: The present study received no financial support.
Acknowledgements: None to be declared.
Authors' contributions: RMA and SOA were significant contributors to the conception of the study and the literature search for related studies. AI, DTG, AHA, BAM and SMA were involved in the literature review, the study's design, and the critical revision of the manuscript, and they participated in data collection. MAG, DAI and HRA were involved in the literature review, study design, and manuscript writing. RJR was the radiologist who performed the assessment of the case. RMA was the pathologist who performed the diagnostic of the case. RMA and MAG confirm the authenticity of all the raw data. All authors approved the final version of the manuscript.
Use of AI: AI was not used in the drafting of the manuscript, the production of graphical elements, or the collection and analysis of data.
Data availability statement: Not applicable.
References
- SSorial V, Khan AS, Welsh T, Zhang L, Fayek SA. Gastric Schwannoma in an Octogenarian: A Case Report and Review of the Literature. Cureus. 2024;16(4). doi:10.7759%2Fcureus.58857.
- Abdullah AS, Ahmed AG, Mohammed SN, Qadir AA, Bapir NM, Fatah GM et al. Benign Tumor Publication in One Year (2022): A Cross-Sectional Study. Barw Medical Journal. 2023;1(4):20-25. doi:10.58742/wefvkv74.
- Majdoubi A, El Achchi A, El Hammouti M, Bouhout T, Serji B. Gastric schwannoma: The gastrointestinal tumor simulator-case report and literature review. International Journal of Surgery Case Reports. 2024; 116:109389. doi:10.1016/j.ijscr.2024.109389.
- Manjesh KA, Nelluri N, Narra AR, Kota SR, Abuji K, Narra AR, et al. Common Benign Nerve Sheath Tumor at an Uncommon Location-Gastric Schwannoma. Cureus. 2024; 16(6). doi:10.7759/cureus.62569.
- Huang S, Huang S, Fang T. Endoscopic full-thickness resection of a large gastric schwannoma and iatrogenic cervical esophageal perforations: A case report.Medicine.2024;103(28):e38808. doi:10.1097/MD.0000000000038808
- Kostovski O, Trajkovski G, Ristovski G, Kunovska SK, Kostovska I. Gastric schwannoma: a case report. Journal of Surgical Case Reports. 2024;2024(3): doi:10.1093/jscr/rjae181 .
- Kakamad FH, Abdalla BA, Abdullah HO, Omar SS, Mohammed SH, Ahmed SM, et al. Lists of predatory journals and publishers: a review for future refinement. European Science Editing. 2024;50:e118119. doi:10.3897/ese.2024.e118119
- Prasad S, Nassar M, Azzam AY, Jose FG, Jamee M, Sliman RKA, et al. CaReL Guidelines: A Consensus-Based Guidelines on Case Reports and Literature Review (CaReL). Barw Medical Journal. 2024;2(2):13-19. doi:10.58742/bmj.v2i2.89.
- Takemura M, Yoshida K, Takii M, Sakurai K, Kanazawa A. Gastric malignant schwannoma presenting with upper gastrointestinal bleeding: a case report. Journal of Medical Case Reports. 2012;6:1-4. doi:10.1186/1752-1947-6-37.
- Kormann S, Semadeni GM, Weber M, Brand S, Borovicka J, Frei R. Combining Endoscopic Submucosa Dissection and Endoscopic Full-Thickness Resection for Resection of a Gastric Schwannoma Mimicking Gastrointestinal Stroma Tumor. ACG Case Reports Journal. 2024;11(7): e01440. doi:10.14309/crj.0000000000001440.
- Lin XM, Liu J, Xi CH, Wang J, Yang GD, Wang XF, Yuan C. Compression from a retroperitoneal schwannoma presenting as a subepithelial lesion of the gastric fundus. Endoscopy. 2024; 56(S 01):E236-7. doi:10.1055/a-2271-4028.
- Huang Y, Huang Y, Jin H, Pei X. Treatment of gastric paralysis after gastric schwannoma by electroacupuncture: A case report and literature review. EXPLORE. 2024 ;20(4): 592-6. doi:10.1016/j.explore.2023.11.009.

This work is licensed under a Creative Commons Attribution 4.0 International License.

