Desmoid-Type Fibromatosis of The Breast: A Case Series
Abstract
Introduction
Desmoid-type fibromatosis (DTF), also called aggressive fibromatosis, is a rare, benign, locally aggressive condition. Mammary DTF originates from fibroblasts and myofibroblasts within the breast tissue, representing 0.2% of all breast tumors. This study aims to present and discuss the clinical presentation and management of seven cases of breast DTF.
Methods
This single-center case series was conducted at the breast clinic of Smart Health Tower in Sulaymaniyah, Iraq. It included all patients diagnosed as breast DTF by histopathological examination, with those lacking complete data excluded. The patients were treated and managed between January 2021 and August 2024.
Results
This study involved seven female patients with a mean age of 35.29 ± 14.29 years. Clinically, six of them (85.71%) presented with a non-tender palpable breast mass, while one patient (14.28%) reported breast pain. Ultrasound revealed hypoechoic lesions in all cases. The average size of the masses was 29.43 ± 17.26 mm. All patients underwent wide local excision of the breast mass. Histopathological examination confirmed the diagnosis of DTF in all cases.
Conclusion
Diagnosing a desmoid tumor of the breast can be difficult, as it can mimic breast carcinoma. The Wide local excision is often the preferred treatment to prevent future recurrences.
Introduction
Desmoid-type fibromatosis (DTF), also known as aggressive fibromatosis, is a rare benign condition that can develop in various body parts, the most common being the extremities, abdominal wall, and intra-abdominal cavity [1]. Mammary DTF arises from fibroblasts and myofibroblasts within the breast tissue. It accounts for 0.2% of all breast tumors [1,2]. Although it does not metastasize, it is known for its local aggressiveness and high recurrence rate [2].
The term "desmoid" was introduced by Mueller in 1838, deriving from the Greek word "desmos," which refers to its tendon-like consistency. However, MacFarlane first described the disease in 1832 [3].
The DTF may occur sporadically or develop following surgical trauma, the implantation of silicone breast implants, or in association with Gardner's syndrome. While most cases of breast fibromatosis are reported in females, it can also occur in males [1]. It commonly affects individuals between 15 and 60 years of age, with the highest incidence occurring in the third and fourth decades of life [4].
Unpredictable and invasive growth patterns characterize the DTF. The tumor often proliferates during the early stages and may also accelerate due to pregnancy or hormonal changes. After this initial growth phase, many patients experience an extended period where the tumor remains stable [5].
Due to their rarity, desmoid tumors present a diagnostic and therapeutic challenge, as they often initially resemble breast carcinoma and have a high tendency for recurrence [6]. This study aims to describe and discuss the presentation and management of seven cases of breast DTF. All the references cited in this study were evaluated for eligibility [7].
Methods
Study design and setting
This single-center case series was conducted at the breast clinic of Smart Health Tower (Sulaymaniyah, Iraq). The patients were treated and managed between January 2021 and August 2024.
Participants
The study included all patients with confirmed breast DTF based on histopathological examination, while those with incomplete data were excluded.
Data collection
Data were collected from the hospital’s registry, including demographic details, clinical presentation, physical examination, medical history, breast ultrasound and mammography findings, core needle biopsy (CNB) and fine needle aspiration cytology (FNAC) results, histopathological examination (HPE), tumor size, magnetic resonance imaging (MRI) and computed tomography (CT) scan findings, types of surgeries performed, postoperative complications, follow-up duration, and recurrence.
Intervention
All patients underwent surgery under general anesthesia with the supine position following skin preparation and disinfection. The procedures involved making elliptical, radial, and semicircular incisions over the breast where the mass was suspected. After the skin was incised, a wide local excision (WLE) of the mass was performed. In one case, the mass extended into the chest wall muscles and involved two ribs, which were also excised. The long thoracic and thoracodorsal nerves were preserved in all cases. Lymph nodes from levels I, II, and III were removed. Hemostasis was achieved, a Redivac drain was inserted for each patient, and a chest tube drain was placed for one patient. The surgical site was then closed in layers.
Histopathological examination and preparation
Regarding pathologic examination, the specimens were fixed in 10% neutral buffered formalin for 24 hours prior to grossing. After that, the specimens were examined systemically, with appropriate sections taken from the tumors and the margins. The resultant blocks were then processed with the Sakura Histo-Tek VP1 automated processor using a standard 11-hour processing protocol through alcohol, xylene, and paraffin. Following embedding in paraffin and trimming, the blocks were sectioned onto regular glass slides, kept in an oven overnight, and then stained manually for hematoxylin and eosin (H&E) using Gill II hematoxylin. The slides were then dried, and coverslips were applied.
For immunohistochemistry, the paraffin blocks were sectioned onto charged glass slides and kept in an oven overnight. Antigen retrieval was achieved through boiling using the Dako PT Link with a solution of pH 6 or 9, depending on the target antibody. The slides were then washed with buffer solution and welled using the Dako Pen, followed by blocking endogenous peroxidase using hydrogen peroxide. The primary antibodies were then applied, followed by the secondary antibody (horseradish peroxidase) and the chromogen (diaminobenzidine). Counterstaining was achieved using hematoxylin Gill II, followed by drying and applying coverslips.
Data analysis
The data were collected using an Excel spreadsheet (Microsoft Excel 2021). They were analyzed qualitatively with Statistical Package for the Social Sciences (SPSS) software (version 27.0) and presented as mean, range, frequencies, and percentages.
Results
This study included seven female patients with an average age of 35.29 ± 14.29 years, ranging from 18 to 61 years. All of the patients were unemployed, and five of them were married (71.43%). Five patients' medical histories were unremarkable (71.43%), while two patients (28.57%) had hypothyroidism. Three patients (42.86%) had previously undergone different types of breast surgeries. None of the patients had a family history of breast cancer.
Among the clinical presentations, six patients (85.71%) had a non-tender palpable breast mass, while one patient (14.28%) experienced breast pain. Ultrasound showed hypoechoic lesions in all patients with BI-RADS 4 or 5 in four patients (57.14%). The average mass size was 29.43 ± 17.26 mm, ranging from 15 mm to 70 mm. In three cases (28.57%), MRI was used, revealing either heterogeneous or homogeneous masses. In one case (14.28%), a CT scan detected a mass in the chest wall with muscle invasion. A CNB was performed in 5 patients (71.43%), and FNAC of the axillary lymph node, which indicated benign lymphoid tissue, was performed in one patient (14.28%).
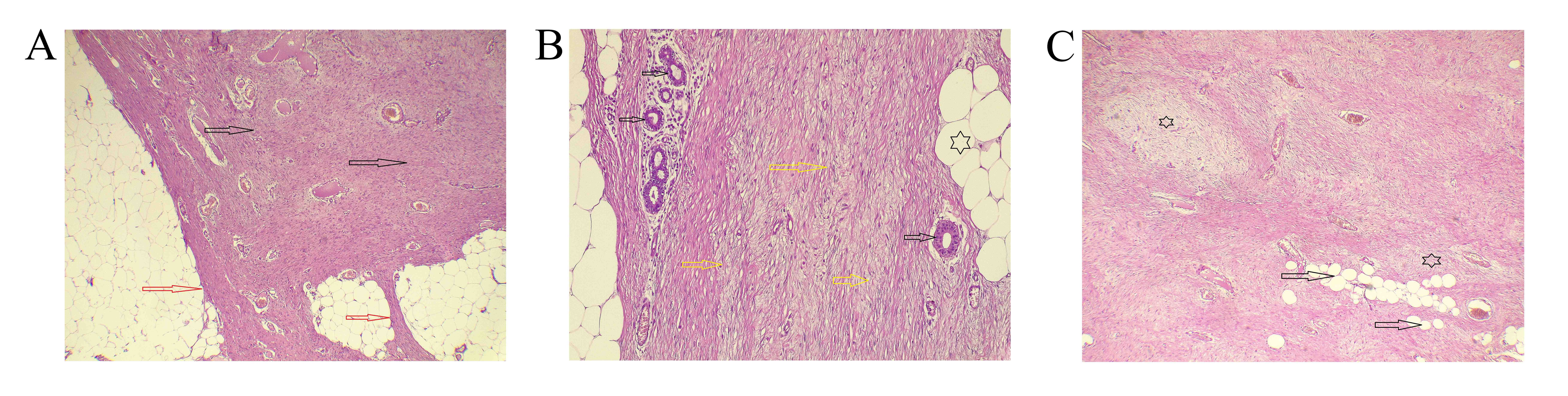
Mammography was performed in only one case (14.28%), showing scattered fibro-glandular density and benign calcification. All patients underwent WLE of the breast mass. In one case (14.28%), surgery required rib excision due to chest wall invasion in a recurrent desmoid tumor. The HPE in all cases, along with immunohistochemistry in 5 cases, confirmed the diagnosis of DTF, with no evidence of lymph node metastasis (Fig. 1). The mean follow-up period was 1.51 ± 0.98 years, ranging from 0.7 to 3 years, with no reported recurrences (Table 1).
| Case # |
Age (year)/Gender |
Occupation | Marital status | Medical history | Surgical history | Presentation | Mass size, mm (US), BI-RADS | MMG, BI-RADS | MRI | CT scan | HPE of CNB | FNAC of axillary LN | Operation | Post op. comp | Follow-up | Rec. |
|
1st |
45/F |
Unemployed |
Married |
Hypothyroidism |
WLE of breast mass |
Rt breast mass |
16 x 26 mm, U2 |
N/A |
N/A |
A 47 x 30 mm irregular mass on the right chest wall invades the pectoralis and contacts the intercostal muscle. |
Fibromatosis |
N/A |
Rt breast mass WLE with the removal of two ribs, achieving clear margins due to a recurrence of fibromatosis after two years. |
No |
2.9 yr |
No |
|
2nd |
61/F |
Unemployed |
Married |
DM, HTN, IHD |
spine surgery, Interventional endoscopy, PCI |
Rt breast mass |
15 x 9 mm, U5 A3 |
fibro glandular density, M2 |
N/A |
N/A |
Fibromatosis |
Benign |
Rt breast mass WLE |
No |
3 yr |
No |
|
3rd |
37/F |
Unemployed |
Married |
Negative |
Rt breast surgery |
Rt breast pain |
42 x 12 mm, U3 |
N/A |
Circumscribed oval mass at 2 o’clock, 35 x 22 mm, with uniform enhancement and heterogeneous fibro glandular tissue. MR-3. |
N/A |
Spindle cell neoplasm |
N/A |
Rt breast mass WLE |
No |
1 yr |
No |
|
4th |
34/F |
Unemployed |
Married |
Negative |
C/S, left breast operation |
Lt breast mass |
70 x 40 mm, U4 |
N/A |
N/A |
Lt breast mass, 75 x 45 mm, invading the 4th to 6th intercostal cartilages and pectoralis major, suggestive of recurrent desmoid tumor. |
Low-grade spindle cell neoplasm, recurrent desmoid type fibromatosis. |
N/A |
Lt breast mass WLE due to fibromatosis recurrence after two years. |
No |
1 yr |
No |
|
5th |
29/F |
Unemployed |
Married |
Negative |
Two C/S |
Lt breast mass |
20 x 10 mm, U3 |
N/A |
Heterogeneous, spiculated enhancement, 45 x 35 mm, with fibro glandular tissue and moderate background enhancement. MR-4. |
N/A |
N/A |
N/A |
Lt breast mass WLE |
No |
1 yr |
No |
|
6th |
24/F |
Unemployed |
Single |
Negative |
Negative |
Rt breast mass |
50 x 20 mm, U4a |
N/A |
N/A |
N/A |
N/A |
N/A |
Rt breast mass WLE |
No |
1 yr |
No |
|
7th |
18/F |
Unemployed |
Single |
Hypothyroidism |
Appendectomy |
Rt breast mass |
70 x 40 mm, U4a |
N/A |
Large circumscribed oval mass, 122 x 90 mm, with heterogeneous enhancement, showing fibro glandular tissue and mild background enhancement. MR-3. |
N/A |
Low-grade spindle cell neoplasm |
N/A |
Rt breast mass WLE |
No |
7 mo |
No |
Discussion
DTF is an intermediate soft tissue tumor marked by clonal fibroblastic proliferation originating in the deep soft tissues. This tumor tends to infiltrate surrounding tissues and has a high local recurrence rate but cannot metastasize [8,9]. They can originate primarily from breast tissue or secondarily from the pectoralis major muscle [1]. This condition typically impacts individuals aged 15 to 60 years, with the highest prevalence observed in the third and fourth decades of life [4]. It is approximately twice as common in women as in men [10]. In this study, the average age of the patients was 35.29 ± 14.29 years, ranging from 18 to 61 years, and all were female.
The exact cause of DTF remains unclear. However, associations with Gardner syndrome, prior trauma, and surgery have been documented [6]. Additionally, while silicone implants are mentioned in the literature as a potential cause, intraoperative trauma is considered more likely to be the primary factor [6].
Clinically, DTF of the breast presents with a wide range of manifestations. It is commonly described as a suspicious, mobile, firm, and painless nodule. However, cases of skin retraction and nipple retraction have also been reported in the literature [6]. Lorenzen et al. described 14 cases of breast DTF in their case series, with all patients presenting with a palpable breast mass [6]. Similarly, in the current study, 85.71% of the patients exhibited a painless breast mass, while only one patient reported breast pain instead of a breast mass.
Relying solely on imaging for diagnosing breast DTF is often inadequate due to its invasive characteristics, which can frequently lead to misdiagnosis as BI-RADS 4 or 5 [10]. On ultrasound, desmoid tumors typically appear as hypoechogenic, ill-defined masses [1]. Mammographically, they present as spiculated masses and, in rare cases, may show calcified deposits [1]. MRI is the most effective imaging technique for assessing the extent of the tumor, particularly when the chest wall is involved [11]. On MRI, desmoid tumors may appear as ill-defined hypointense or isointense masses on T1-weighted images and hyperintense masses on T2-weighted images [12]. In the present study, ultrasound showed hypoechoic lesions in each patient, and ultrasound showed BI-RADS 4 or 5 in 57.14%. Mammography was performed for one patient, showing scattered fibro-glandular density with benign calcifications. MRI was done for three patients, revealing either homogeneous or heterogeneous enhancing masses and varying sizes of fibro-glandular tissue. Additionally, CT scans of one case showed a chest wall mass with invasion of the pectoralis muscle.
Diagnosing a desmoid tumor of the breast can be challenging both clinically and radiologically, as it may resemble breast carcinoma [1]. A definitive diagnosis is only possible through HPE of the lesion. The tumor appears macroscopically as a grayish-white, firm, irregular nodular mass [1]. Microscopically, it is characterized by spindle cells with varying amounts of collagen fiber deposition [12]. Histopathologically, desmoid tumors must be distinguished from scar tissue, fibrosarcoma, or fibromatosis-like metaplastic spindle cell tumors [12].
Immunohistochemical analysis, particularly of preoperative biopsies, can aid in diagnosing desmoid fibromatosis. β-Catenin is a key immunohistochemical marker, showing nuclear expression in sporadic and familial cases in up to 80% of patients [6]. Mutations in the β-catenin gene (CTNNB1) are found in most cases of sporadic DTF, leading to the accumulation of this oncoprotein in the nucleus [6]. Elevated β-catenin levels activate the WnT signaling pathway, contributing to tumor development [6]. However, nuclear β-catenin staining is not specific to breast desmoid fibromatosis; it is also occasionally observed in spindle cell carcinomas, 23% of metaplastic breast carcinomas, and up to 93% of benign phyllodes tumors [13].
The treatment of breast desmoid tumors remains controversial due to the limited data available, given the low incidence of the disease [14]. Various approaches may be employed, including surgery, radiotherapy, chemotherapy, and hormonal therapy. Radical surgical excision is generally considered the treatment of choice [1]. However, complete excision may be avoided in carefully selected patients, especially when the surgery could result in poor functional or cosmetic outcomes, such as in cases where the lesion is superficial or subareolar and may require the removal of the nipple-areolar complex [14]. For some women, mastectomy may be recommended if they experience multiple recurrences, have a large tumor, or face difficulties in obtaining a histological diagnosis [15]. This study chose a comprehensive WLE as a preventive measure to avoid any potential future recurrences.
Breast DTF recurrence is common, with a prevalence of 18-29% over 3-6 years. There is also a risk of involvement with thoracic muscles and ribs [15]. WLE can help prevent or reduce the risk of recurrence [1]. For tumors that are unresectable or require extensive surgical procedures, such as major chest wall resection, radiotherapy may be considered as an alternative treatment option [16]. In this study, no recurrences were observed during the follow-up period. The mean follow-up duration was 1.51 ± 0.98 years, ranging from 0.7 to 3 years.
Systemic therapy can be employed for unresectable or recurrent desmoid tumors, as well as for patients who cannot tolerate surgical treatment or when the tumor involves the chest wall [1]. This therapy includes non-cytotoxic approaches, such as hormonal therapy with Tamoxifen alone or in combination with nonsteroidal anti-inflammatory drugs, as well as cytotoxic approaches like chemotherapy [1]. In the current study, one patient underwent a WLE and the removal of two ribs due to an irregular mass on the right chest wall that invaded the pectoralis muscle and contacted the intercostal muscle. A limitation of this study is the short follow-up duration, which may not be long enough to identify potential recurrences.
Conclusion
Diagnosing a desmoid tumor of the breast can be difficult, as it can mimic breast carcinoma. The WLE is often the preferred treatment to prevent future recurrences.
Declarations
Conflicts of interest: The author(s) have no conflicts of interest to disclose.
Ethical approval: The study's ethical approval was obtained from the scientific committee of the Kscien Organization for Scientific Research.
Patient consent (participation and publication): Verbal informed consent was obtained from patients for publication.
Source of Funding: Smart Health Tower
Role of Funder: The funder remained independent, refraining from involvement in data collection, analysis, or result formulation, ensuring unbiased research free from external influence.
Acknowledgements: None to be declared.
Authors' contributions: AMS and AMA were major contributors to the conception of the study, as well as to the literature search for related studies. HAN, MKA and AAQ were involved in the literature review, study design, and writing the manuscript. ZDH, HAY, SHH, SOK, FHK and HAS were involved in the literature review, the design of the study, the critical revision of the manuscript and the processing of the figures. FHK and AMS confirm the authenticity of all the raw data. RMA was the pathologist who performed the histopathological diagnosis. LRAP was the radiologist who performed the assessment of the case. All authors have read and approved the final manuscript.
Use of AI: AI was not used in the drafting of the manuscript, the production of graphical elements, or the collection and analysis of data.
Data availability statement: Note applicable.
References
- Pankratjevaitė L, Ceslevičienė I, Poškienė L, Boguševičius A. A case report of desmoid type fibromatosis of the breast. Lietuvos chirurgija. 2019;18(2):107-11. doi:10.15388/lietchirur.2019.18.12
- Benej R, Mečiarová I, Pohlodek K. Desmoid-type fibromatosis of the breast: A report of 2 cases. Oncology letters. 2017;14(2):1433-8. doi:N/A
- Garcia-Ortega DY, Martín-Tellez KS, Cuellar-Hubbe M, Martínez-Said H, Álvarez-Cano A, Brener-Chaoul M et al. Desmoid-type fibromatosis. Cancers. 2020;12(7):1851. doi:10.3390/cancers12071851
- Braschi-Amirfarzan M, Keraliya AR, Krajewski KM, Tirumani SH, Shinagare AB, Hornick JL et al. Role of imaging in management of desmoid-type fibromatosis: a primer for radiologists. Radiographics. 2016;36(3):767-82. doi:10.1007/s13244-015-0427-8
- Timbergen MJ, Schut AR, Grünhagen DJ, Sleijfer S, Verhoef C. Active surveillance in desmoid-type fibromatosis: a systematic literature review. European Journal of Cancer. 2020;137:18-29. doi:10.1016/j.ejca.2020.06.022
- Lorenzen J, Cramer M, Buck N, Friedrichs K, Graubner K, Lühr CS et al. Desmoid type fibromatosis of the breast: ten-year institutional results of imaging, histopathology, and surgery. Breast Care. 2021 Feb 16;16(1):77-84. doi:10.1159/000507842
- Abdullah HO, Abdalla BA, Kakamad FH, Ahmed JO, Baba HO, Hassan MN, et al. Predatory Publishing Lists: A Review on the Ongoing Battle Against Fraudulent Actions. Barw Medical Journal. 2024;2(2):26-30. doi:10.58742/bmj.v2i4.138
- Abdullah AS, Ahmed AG, Mohammed SN, Qadir AA, Bapir NM, Fatah GM et al. Benign Tumor Publication in One Year (2022): A Cross-Sectional Study. Barw Medical Journal. 2023;1(4):20-25. doi:10.58742/wefvkv74
- Eastley N, McCulloch T, Esler C, Hennig I, Fairbairn J, Gronchi A et al. Extra-abdominal desmoid fibromatosis: a review of management, current guidance and unanswered questions. European Journal of Surgical Oncology (EJSO). 2016;42(7):1071-83. doi:10.1016/j.ejso.2016.02.012
- Hennuy C, Defrère P, Maweja S, Thiry A, Gennigens C. Bilateral breast desmoid-type fibromatosis, case report and literature review. Gland Surgery. 2022;11(11):1832. doi:10.21037/gs-22-271
- Park AR, Kim JY, Kim HJ, Kim JH, Jeong MJ, Kim SH et al. Slow growing desmoid-type fibromatosis of the breast: a case report. Journal of the Korean Society of Radiology. 2017;76(4):259-63. doi:10.1055/s-0036-1578716
- Erguvan-Dogan B, Dempsey PJ, Ayyar G, Gilcrease MZ. Primary desmoid tumor (extraabdominal fibromatosis) of the breast. AJR. American Journal of roentgenology. 2005;185(2):488-9. doi:10.2214/ajr.185.2.01850488
- Lacroix-Triki M, Geyer FC, Lambros MB, Savage K, Ellis IO, Lee AH, et al. β-catenin/Wnt signalling pathway in fibromatosis, metaplastic carcinomas and phyllodes tumours of the breast. Modern Pathology. 2010;23(11):1438-48. doi:10.1002/path.2763
- Wongmaneerung P, Somwangprasert A, Watcharachan K, Ditsatham C. Bilateral desmoid tumor of the breast: case seriesand literature review. International Medical Case Reports Journal. 2016:247-51. doi:10.2147/imcrj.s106325
- Bouab M, Harit A, Boufettal H, Mahdaoui S, Samouh N. Desmoid fibromatosis of the breast occurring after breast reduction surgery mimicking a carcinoma: A rare case report. Annals of Medicine and Surgery. 2022;77:103526. doi:10.1016/j.amsu.2022.103526
- Ha KY, DeLeon P, Hamilton R. Breast fibromatosis mimicking breast carcinoma. In Baylor University Medical Center Proceedings 2013;26 (1);22-24. Taylor & Francis. doi:N/A

This work is licensed under a Creative Commons Attribution 4.0 International License.

